Pattern of resistance on first-line EGFR-directed therapy in EGFR-positive metastatic NSCLC
Sree Siva Kumar Raja Addagalla, Vanita Noronha, Nandini Menon, Minit Shah, Amit Joshi, Anokhi Shah and Kumar Prabhash
Department of Medical Oncology, Tata Memorial Centre, Homi Bhabha National Institute (HBNI), Mumbai 400094, India
https://orcid.org/0009-0009-0042-8609
https://orcid.org/0000-0002-4214-5799
https://orcid.org/0009-0004-3769-3194
https://orcid.org/0000-0001-8858-5004
Abstract
Background and purpose: Resistance to epithelial growth factor receptor gene (EGFR)-tyrosine kinase inhibitors, both intrinsic and acquired, presents a major challenge in EGFR mutant non-small cell lung carcinoma (NSCLC), with the T790M mutation being the most common acquired resistance mechanism. Data on resistance patterns in the Indian population remains limited.
Materials and methods: This post hoc analysis of a Phase III trial conducted at Tata Memorial Centre (TMC) in Mumbai, India, included 350 patients with advanced EGFR-mutant NSCLC. Patients were randomised to receive either gefitinib alone (n = 176) or gefitinib with chemotherapy (pemetrexed and carboplatin, n = 174). The primary objective was to identify resistance mechanisms following progression. Secondary objectives included comparing resistance patterns between the two arms and assessing progression rates and outcomes. At progression, histological and molecular evaluation was done with reverse transcription polymerase chain reaction (RT-PCR), anaplastic lymphoma kinase – immunohistochemistry (ALK IHC) and/or next generation sequencing (NGS) at physician discretion.
Results: Of the 275 (78.6%) patients experiencing progressive disease (PD), a total of 206 patients were available for the final analysis after excluding patients without histological/molecular analysis.
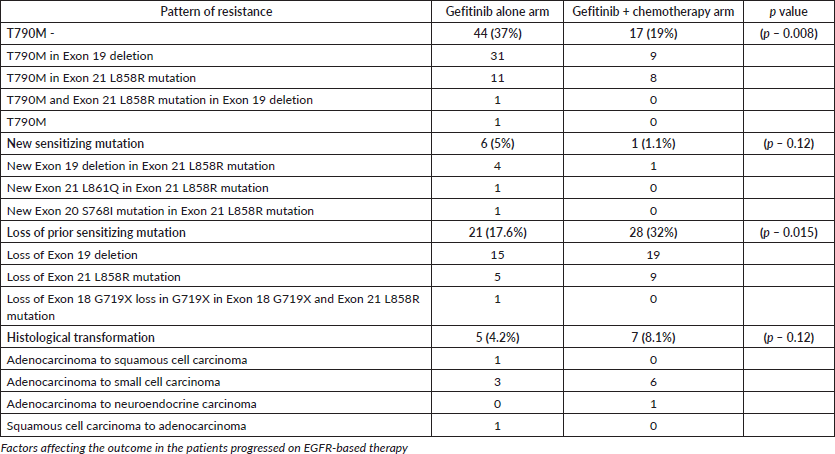
Histological transformation to small cell lung carcinoma (SCLC) occurred in 12 patients (6%), with various EGFR mutation statuses identified. T790M mutations were observed in 44 (37%) of 119 patients in the gefitinib arm and 17 (19%) of 87 patients in the gefitinib plus chemotherapy arm (p = 0.008). A new sensitising mutation was found in 6 (5%) in the gefitinib arm and in 1 (1.1%) in the gefitinib plus chemotherapy arm (p – 0.12). Loss of prior sensitising mutation was found in 21 (17.6%) in the gefitinib arm and in 28 (32%) in the gefitinib plus chemotherapy arm (p – 0.015).
Patients with T790M mutations had a progression-free survival 2 of 22.7 months (95% CI: 19.4–27.4 months), compared to 19.2 months (95% CI: 17.5–22.9 months) in those
without T790M mutations (p = 0.95). Overall survival was 27.9 months (95% CI: 24.6–34.6 months) in the T790M group compared to 26.5 months (95% CI: 23.2–30.1 months) in the non-T790M group (p = 0.75).
Interpretation: Emergence of T790M was lower than reported in previous studies, likely due to the addition of chemotherapy to gefitinib. T790M mutations were more prevalent in the gefitinib-alone arm. Histological transformation and loss of sensitising mutations highlight the importance of repeat biopsy and molecular testing to guide subsequent treatment decisions.
Keywords: resistance, EGFR-TKIs, NSCLC, T790M, chemotherapy
Correspondence to: Kumar Prabhash.
Email: kumarprabhashtmh@gmail.com
Published: 09/03/2026
Received: 02/08/2025
Publication costs for this article were supported by ecancer (UK Charity number 1176307).
Copyright: © the authors; licensee ecancermedicalscience. This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
The identification of driver mutations like epithelial growth factor receptor gene (EGFR), ALK and subsequent development of effective tyrosine kinase inhibitors (TKIs) against them has transformed therapy in metastatic non-small cell lung carcinoma (NSCLC) [1]. The incidence of EGFR-mutated NSCLC was found to be about 45.8% in the Indian population [2]. EGFR is a tyrosine kinase receptor which binds to its ligand epidermal growth factor, leading to receptor dimerisation, auto-phosphorylation at the tyrosine kinase residues, activating cellular signaling pathways such as the phosphoinositide 3-kinase-protein kinase B pathway, the signal transducer and activator of transcription pathway and the mitogen-activated protein kinase pathway, resulting in increased cell proliferation, migration and survival [3–5]. EGFR-activating gene mutations cause constitutive EGFR activation, which leads to cell proliferation. The most common mutations occur in exons 19 and 21 accounting for approximately 45% and 40% of all EGFR mutations, respectively, and are associated with good responses to EGFR-targeted small molecule inhibitor therapies [5]. Less common mutations include G719X in exon 18, L861Q in exon 21, and insertions in exon 20 (Ex20ins) are each estimated to constitute about 2% of EGFR mutations [6–8].
Initial studies in EGFR mutant NSCLC have shown better outcomes with the first and second generation TKIs compared to chemotherapy [9–15]. In the Indian setting, our team at Tata Memorial Hospital, (Mumbai, India) showed that combination of chemotherapy with the first generation TKI (gefitinib) improved survival [16]. Subsequently, the third generation EGFR-TKI, osimertinib, was introduced, which was active against the T790M resistance mutation [17]. It was also shown to be efficacious in the first line setting, with a superior overall survival (OS) and progression free survival (PFS) as compared to first generation EGFR TKIs as demonstrated in the FLAURA trial [17, 18]. In the FLAURA 2 trial, osimertinib plus chemotherapy (pemetrexed with cisplatin or carboplatin) was compared against osimertinib and this showed a PFS benefit with chemotherapy (25.5 versus 16.7 months , HR 0.62; p < 0.01) and at 2 years, the OS was not reached versus 36.7 months (HR, 0.75; 95% CI, 0.57–0.97; p = 0.0280 with an overall maturity of 41%) with increased hematological toxicity [19]. In the MARIPOSA trial, amivantamab with lazertinib improved median PFS (23.7 versus 16.6 months, HR 0.7 p < 0.001) and improved interim OS (HR 0.8) when compared to osimertinib [20]. Despite superior outcomes of these newer agents, high cost and less availability limit their use in routine clinical practice. Osimertinib costs about 3.2 lakhs INR per month. Amivantamab costs about 10 lakhs INR in the first month and 5 lakhs per month in the subsequent months and lazertinib is currently not marketed in India. This situation led to the use of early generation TKIs alone or in combination with chemotherapy. But after an initial response, they invariably lead to resistance.
Resistance to EGFR TKIs can be classified into two major categories: intrinsic and acquired. Intrinsic resistance accounts for 20%–30% of cases and is related to poor initial response to TKIs. It can be attributed to pre-existing de novo EGFR-dependent or independent mechanisms, whereas the acquired resistance can arise under therapeutic selective pressure, as a result of either the expansion of pre-existing sub-clonal populations or the evolution of drug-tolerant cells [20]. Different mechanisms of acquired resistance to first-generation EGFR-TKIs have been reported [21]. EGFR T790M mutation is the most common mechanism occurring in 50%–60% of cases [21–24]. Although, a number of studies have been done to elucidate the patterns of resistance in the patients who have progressed on EGFR-TKIs, such data in Indian population is sparse and remains an unmet need.
Materials and methods
General study details
This was a post hoc analysis of the randomised, open-label, phase III study that compared gefitinib with gefitinib plus chemotherapy in patients with advanced NSCLC with activating EGFR mutations in the first-line setting. The methodology has been published in detail already [16]. The trial was conducted at TMC, an academic tertiary oncology hospital in Mumbai, India. A total of 350 patients were enrolled in the study. The study was approved by the Institutional Ethics Committee of TMC and monitored by the Data Safety Monitoring Subcommittee. All patients provided written informed consent. The trial was conducted according to the principles laid down by the International Conference on Harmonisation Good Clinical Practice guidelines, the Declaration of Helsinki, and Schedule Y (Drugs and Cosmetic Act, 1940) and the guidelines established by the Indian Council of Medical Research. The trial was registered at Clinical Trials Registry–India (identifier: CTRI/ 2016/08/007149).
Participants
The patients were randomised to receive either Gefitinib alone or Gefitinib along with chemotherapy. Patients who had progressed underwent re-biopsy, depending on feasibility.
Study aim/objective
Our primary objective was to describe the mechanisms of resistance in patients who progressed on either gefitinib alone or in combination with chemotherapy. The secondary objectives included the differences in the resistance patterns between patients who progressed on gefitinib alone versus those who progressed on gefitinib along with chemotherapy, the patterns of progression and progression rates among various EGFR mutations and factors affecting outcomes.
Study methodology
At progression (clinical or radiological), all the patients were evaluated for the feasibility of re-biopsy. In patients in whom biopsy was not possible, if any fluid collection was present – cytology and cell block were obtained. All the patients were evaluated for the feasibility of liquid biopsy on blood and this was done whenever possible. We excluded the patients in whom neither tissue biopsy nor liquid biopsy were possible.
In the patients for whom either biopsy or cell block was available, histopathological evaluation and EGFR RT-PCR was done in all and ALK IHC and NGS was done in some of the patients when possible and affordable based on the physician discretion.
Stastistics
Using descriptive statistical analysis, the patterns of progression, mechanisms of resistance and variations in their patterns between the gefitinib and gefitinib plus chemotherapy groups were computed. The difference in the emergence of T790M mutation between the two arms were compared using the Pearson chi-square test and a ‘p’ value of < 0.05 was considered as significant. Progression free survival 2 (PFS2) was calculated from the date of randomisation to either the date of second progression or date of last follow-up or date of death if there is no event of second progression using Kaplan Meir analysis with a ‘p’ value of < 0.05 being considered as significant. The event for PFS2 was taken as either second progression or death. OS was calculated from the date of randomisation to the date of death or date of last follow-up using Kaplan Meir analysis with a ‘p’ value of < 0.05 being considered as significant. Death was taken as the event for OS. Patients who were lost to follow-up were censored while calculating both OS and PFS2.
Results
Patient predisposition
350 patients who were enrolled between August 2016 and 2018 and 176 (50.3%) patients were assigned to gefitinib and 174 (49.7%) patients were assigned to Gefitinib along with pemetrexed and carboplatin. These patients were followed up over the period till May 2024.
Baseline characteristics
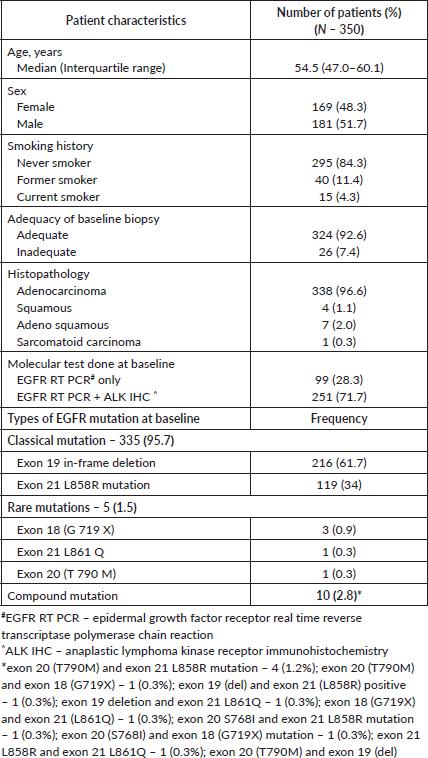
The patients’ baseline characteristics with respect to demographic data and the molecular data are listed in Table 1 as follows.
The outcomes were tabulated in the Supplementary Table 1, and the PFS among various molecular mutations were tabulated in Supplementary Table 2. The consort diagram was in Figure 1. Of the 275 patients who had PD (the sites of progression were tabulated in Table 2), 10 (3.7%) had subjective PD alone (imaging could not be performed for various reasons) and 265 (96.3%) were noted to have radiological PD. Of the 265, 110 (40%) had radiological PD alone and 155 (56.3%) had both radiological and subjective PD. The details of the sites of biopsy were tabulated in the Supplementary Table 4, while the complications were tabulated in Supplementary Table 5.
Tissue biopsies were obtained on 157 (57.1%) patients and a cell block in 14 (5.1%). Liquid biopsy was performed in 169 (61.5%) patients. The details of repeat biopsy given in the Table 3.
Of the nine patients who had histological transformation to small cell carcinoma, the results of molecular testing were 6 had Exon 19 retained, 1 had loss of exon 19, 1 had new mutation of S768I along with L858R retained, 1 had inadequate biopsy for molecular testing.
Resistance patterns – gefitinib versus gefitinib plus chemotherapy
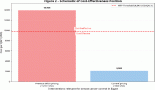
The molecular analysis of the patients at progression were tabulated in Supplementary Tables 6 and 7. Additional molecular analysis noted on NGS were tabulated in Supplementary Table 8. T790M has been noted in 44 (37%) out of 119 patients in the gefitinib arm, whereas it was noted in 17 (19%) out of 87 patients in the gefitinib plus chemotherapy arm (p – 0.008) Figure 2. Patients receiving chemotherapy plus gefitinib, SCLC transformation was found in 6 out of 87(7%), while it was found in 3 out of 119 patients (2%) in the gefitinib arm (p - 0.12). The comparison between the two groups is charted in Table 2, and the changes in the overall cohort and individual arms are depicted in Figure 3.
T790M status
PFS2 among the patients with acquired T790M was 22.7 months (95% CI – 19.4 – 27.4 months), while the PFS 2 in patients without T790M was 19.2 months (95% CI – 17.5 – 22.9 months) (p = 0.95). Majority of them received osimertinib at progression (48 out of 57(84.2%) patients with T790M). The OS was 27.9 months (95% CI – 24.6 – 34.6 months) in the patients with T790M compared to 26.5 months (95% CI – 23.2 – 30.1 months) in in patients without T790M (p – 0.75). The survival curves are provided in Supplementary Data Figures 1 and 2.
Site of progression
PFS2 among the patients with central nervous system (CNS) progression was 22.1 months (95% CI – 19.1 – 30.9 months), while the PFS 2 in patients progressed at other sites was 20.0 months (95% CI – 18.3 – 23.3 months) (p = 0.38). The OS was 27.2 months (95% CI – 24.9 – 31.3 months) in patients with CNS progression compared to 22.2 months (95% CI – 19.0 – 30.1 months) in the patients progressed other sites (p – 0.57). The survival curves are provided in Supplementary Data Figures 3 and 4.
Table 1. Baseline demographic and molecular characteristics.
Outcomes of patients with other resistance patterns
Patients with SCLC transformation received platinum plus etoposide combination along with continuation of TKI and all of them initially responded but later had a progression with a median OS from the point of transformation being 14.2 months. In patients who had new sensitising mutations, the continuation of gefitinib (with one patient being started on osimertinib in view of progression in brain) and chemotherapy added if rapid response was needed, yielded an addition PFS2 of nearly 8 months in this group. One patient having ALK mutation was started on crizotinib and it has given a progression-free interval of 3.3 years in that patient.
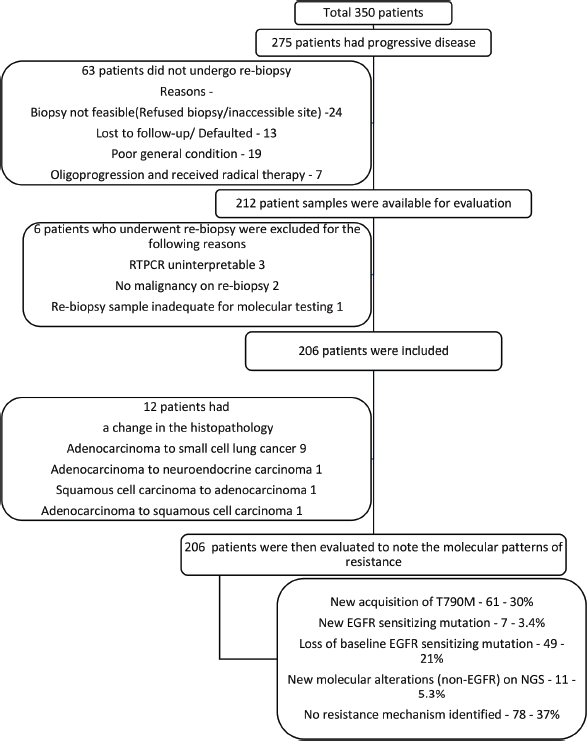
Figure 1. Consort diagram.
Table 2. Comparison of resistance pattern between the treatment groups.
Discussion
In our study, in patients with EGFR mutant NSCLC treated with either gefitinib or gefitinib plus chemotherapy in the first line setting, the most common pattern of resistance was T790M mutation noted in 30% patients. In the literature, prior studies also have shown the predominant pattern of resistance as T790M in exon 20, being 49% in the study by Sequist et al [22] 62% in the study by Yu et al [23] and 58% in the IMPRESS trial [25]. The emergence of T790M in the gefitinib alone arm (37%) was relatively similar to these studies, which also used TKI alone. The emergence of T790M as a pattern of resistance was significantly more in the gefitinib alone arm (37%) than the gefitinib plus chemotherapy arm (19%). This phenomenon of suppression of the emergence of the T790M clone in the arm receiving chemotherapy has not been previously documented.
Among the studies that compared TKI to TKI plus chemotherapy, NEJ009 (gefitinib plus chemotherapy versus gefitinib alone) did not report the resistance patterns, in IMPRESS trial (gefitinib plus chemotherapy versus placebo plus chemotherapy), the T790M was noted in 62% of the patients without any difference between the two arms. The comparison was also studied in the preliminary data published in the FLAURA 2 trial (osimertinib versus osimertinib plus chemotherapy) [28]. The patterns noted in the FLAURA 2 were not significantly difference between the two arms, although numerically higher EGFR C797S was observed in the osimertinib monotherapy arm. The resistance patterns observed were mesenchymal epithelial transition factor (MET) amplification (11.9%), EGFR C797S (8.7%), uncommon EGFR mutations (2.3%), Phosphatidylinositol – 4,5 – bisphosphate 3-kinase catalytic subunit alpha (PIK3CA) (6.3%), CDK 4/6 amplification (5.5%), Cyclin D/E amplification (3.2%), human epidermal growth factor receptor 2 (HER2) amplification (3.2%), v-raf murine sarcoma viral oncogene homolog B (BRAF) V600E (3.2%), rearranged during transfection fusion (3.2%), BRAF fusion (3.2%), ALK fusion (1.6%), other fusions (5.5%) and no known resistance pattern noted in 42% patients [28]. These patterns were different from our study as osimertinib is active against the T790M and it was, therefore, not noted in the FLAURA 2 study. The other changes being less frequently noted might be due to the less frequent use of NGS in our study.
Table 3. Details of results of repeat biopsy in patients who had PD.
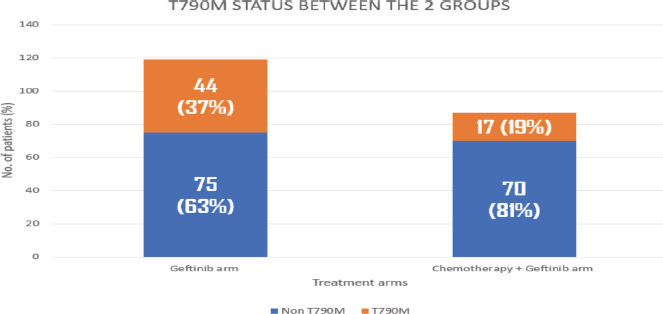
Figure 2. Histogram showing comparison of T790M in geftinib versus geftinib + chemotherapy arms.
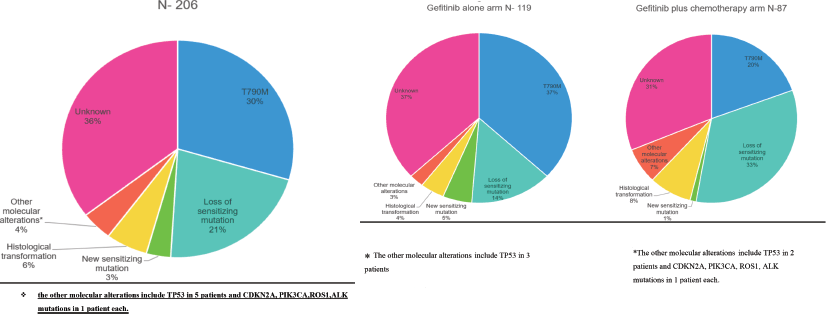
Figure 3. (a): Overall resistance mechanisms in the study population. (b): Resistance mechanisms in the gefitinib arm. (c): Resistance mechanisms in the gefitinib plus chemotherapy.
The lower rates of T790M observed in the present study, might be due to the fact that patients in one arm received chemotherapy along with TKI and being a biologically different population. Among the patients with T790M and those with CNS progression in whom the EGFR sensitising mutation was retained, 62 patients received osimertinib in the 2nd line and they had a PFS of 23.3 months compared to 18.9 months in the rest of the cohort (p – 0.28). These data were similar to the APPLE study, where osimertinib was used sequentially after emergence of T790M in circulating tumour DNA on gefitinib (22.1 months in osimertinib upfront versus 21.9 months in the sequential arm) [26]. This provides the rationale for sequential therapy, especially in resource constrained settings.
Among the other mechanisms of resistance noted in our study was loss of prior sensitising mutation, seen in 21%, which was noted in 27% in a smaller study done by Tabara et al [27]. It was also noted in a case report [28]. This loss of prior sensitising mutations was higher in the chemotherapy plus gefitinib arm (32%) compared to the gefitinib alone arm (17%) (p – 0.015). This pattern of loss of sensitising mutation more in the TKI plus chemotherapy treated patients has not been previously documented. This might be due to the possible conversion to a non-molecularly driven tumour implications for subsequent lines of therapy.
Histological transformation was seen in 6% (12 patients), of which nine had a transformation from adenocarcinoma to SCLC. Overall, in the literature transformation has been reported to be between 3% and 10% [22, 23]. The rate of SCLC transformation was not significantly different between the two treatment groups. Patients with SCLC transformation received platinum plus etoposide combination along with continuation of TKI and all of them initially responded but later had a progression with a median OS from the point of transformation being 14.2 months. It has been noted as 10.9 months in the study by Marcoux et al [29]. Patients with transformed SCLC in the setting of EGFR mutant NSCLC appear to have relatively good prognosis as compared to de novo SCLC.
New sensitising mutations were noted in 3% cases. This number was similar to the previous studies [22, 23]. This number was noted to be 4% in the FLAURA 2 trial [30]. In our study, these patients were continued on gefitinib (with one patient being started on osimertinib in view of progression in brain) and chemotherapy added if rapid response was needed. This has given an additional PFS2 of nearly 8 months.
Molecular mutations other than EGFR were noted but the baseline NGS was not performed. But these alterations were known mechanisms of resistance. Tumour protein 53 (TP53) mutation was found in 2% of the cases. The patients having TP53 mutations were known to increased histological transformation rate and early progression and acquired resistance of EGFR based therapy [31]. In our study, we noted 2 cases of SCLC transformation having TP53 mutations. The other changes include – PIK3CA mutation, ALK mutation, cyclin-dependent kinase inhibitor 2A (CDKN2A) mutation and ROS mutation being noted in 1% each. PIK3CA mutation was noted in 5% cases in the study by Sequist et al [22] and 6% in the FLAURA 2 study. ALK mutation was noted after progression on second-line osimertinib; in this case, the addition of crizotinib to osimertinib led to stabilisation of disease [32]. In our study, also crizotinib addition was also done in one patient and has shown an excellent progression free interval of about 3.3 years. CDKN2A was noted in one of the studies as a resistance mechanism post 2nd line osimertinib, but alterations in the wider group of cell cycle regulators were noted in 10%–12% cases [33, 34]. ROS fusion was noted previously in one case report where addition of crizotinib has led to a partial response [35]. The other mechanisms noted in previous studies which we did not find in our cohort were - MET amplification in about 3%–5% cases, HER2 amplification in about 3% cases, PIK3CA mutation in about 1%–5% cases and Beta catenin amplification in 2%–5% cases.
No new pattern of resistance was noted in 36% of the cases. This was in line with what was seen in the previous studies, i.e., 30%–40% [22, 23]. 30% in the study by Yu et al [23] and 40% in the study by Sequist et al [22] Modern analysis like NGS were not done in all the available patients in these studies due to lack of samples or feasibility, which may be the reason for not identifying molecular cause of resistance.
The limitations of our study include the lack of use of NGS which was expensive and not affordable by majority of patients at the study period might have led to missing the patterns of resistance in some patients. About 22% of patients were not included in the final analysis as the repeat biopsy was not feasible. Some patients in this group had early death post progression and the analysis in this group might have helped to note the molecular changes that led poor outcomes.
Conclusion
Our study describes the patterns of resistance in EGFR mutant NSCLC and will help guide the future therapeutic options. Patients with SCLC transformation, T790M mutation, ALK mutation and ROS fusion had meaningful outcomes with molecular therapies post progression. Our study emphasises the need to perform repeat biopsy and molecular analysis at PD.
List of abbreviations
ALK IHC, Anaplastic lymphoma kinase – immunohistochemistry; BRAF gene, v-raf murine sarcoma viral oncogene homolog B; CDKN2A gene, Cyclin-dependent kinase inhibitor 2A; cfDNA, Circulating free deoxyribonucleic acid; CNS, Central nervous system; EGFR, Epithelial growth factor receptor gene; HER2, Human epidermal growth factor receptor 2; NGS, Next generation sequencing; NSCLC, Non-small cell lung cancer; OS, Overall survival; PD, Progressive disease; PFS, Progression free survival; PFS 2, Progression free survival 2; PIK3CA gene, Phosphatidylinositol – 4,5 – bisphosphate 3- kinase catalytic subunit alpha; RT-PCR, Reverse transcription polymerase chain reaction; SCLC, Small cell lung carcinoma; TKIs, Tyrosine kinase inhibitors; TMC, Tata Memorial Center; TP53, Tumour protein 53.
Acknowledgment
We acknowledge the contributions of the thoracic oncology team and data management staff at Tata Memorial Center.
Conflicts of interest
None declared.
Funding
No external funding was received for this study.
References
1. Pao W and Chmielecki J (2010) Rational, biologically based treatment of EGFR-mutant non-small-cell lung cancer Nat Rev Cancer 10(11) 760–774 https://doi.org/10.1038/nrc2947 PMID: 20966921 PMCID: 3072803
2. Jha P, Joshi A, and Mishra R, et al (2024) Landscape of clinically relevant genomic alterations in the Indian non-small cell lung cancer patients Clin Lung Cancer 25(8) e420–e430 https://doi.org/10.1016/j.cllc.2024.07.011 PMID: 39129089
3. Wu YL, Zhou C, and Hu CP, et al (2014) Afatinib versus cisplatin plus gemcitabine for first-line treatment of Asian patients with advanced non-small-cell lung cancer harboring EGFR mutations (LUX-Lung 6): an open-label, randomized phase 3 trial Lancet Oncol 15(2) 213–222 https://doi.org/10.1016/S1470-2045(13)70604-1 PMID: 24439929
4. Sharma SV, Bell DW, and Settleman J, et al (2007) Epidermal growth factor receptor mutations in lung cancer Nat Rev Cancer 7(3) 169–181 https://doi.org/10.1038/nrc2088 PMID: 17318210
5. Lynch TJ, Bell DW, and Sorella R, et al (2004) Activating mutations in the epidermal growth factor receptor underlying responsiveness of non-small-cell lung cancer to gefitinib N Engl J Med 350(21) 2129–2139 https://doi.org/10.1056/NEJMoa040938 PMID: 15118073
6. Paez JG, Janne PA, and Lee JC, et al (2004) EGFR mutations in lung cancer: correlation with clinical response to gefitinib therapy Science 304(5676) 1497–1500 https://doi.org/10.1126/science.1099314 PMID: 15118125
7. Pao W, Miller V, and Zakowski M, et al (2004) EGF receptor gene mutations are common in lung cancers from “never smokers” and are associated with sensitivity of tumors to gefitinib and erlotinib Proc Natl Acad Sci USA 101(36) 13306–13311 https://doi.org/10.1073/pnas.0405220101 PMID: 15329413 PMCID: 516528
8. Kate S, Chougule A, and Joshi A, et al (2019) Outcome of uncommon EGFR mutation positive newly diagnosed advanced non-small cell lung cancer patients: a single center retrospective analysis Lung Cancer (Auckl) 10 1–10 PMID: 30774491 PMCID: 6357894
9. Mok TS, Wu YL, and Thongprasert S, et al (2009) Gefitinib or carboplatin-paclitaxel in pulmonary adenocarcinoma N Engl J Med 361(10) 947–957 https://doi.org/10.1056/NEJMoa0810699 PMID: 19692680
10. Mitsudomi T, Morita S, and Yatabe Y, et al (2010) Gefitinib versus cisplatin plus docetaxel in patients with non-small-cell lung cancer harboring mutations of the epidermal growth factor receptor (WJTOG3405): an open label, randomized phase 3 trial Lancet Oncol 11(2) 121–128 https://doi.org/10.1016/S1470-2045(09)70364-X
11. Maemondo M, Inoue A, and Kobayashi K, et al (2010) Gefitinib or chemotherapy for non-small-cell lung cancer with mutated EGFR N Engl J Med 362(25) 2380–2388 https://doi.org/10.1056/NEJMoa0909530 PMID: 20573926
12. Zhou C, Wu YL, and Chen G, et al (2011) Erlotinib versus chemotherapy as first-line treatment for patients with advanced EGFR mutation-positive non-small-cell lung cancer (OPTIMAL, CTONG-0802): a multicenter, open-label, randomized, phase 3 study Lancet Oncol 12(8) 735–742 https://doi.org/10.1016/S1470-2045(11)70184-X PMID: 21783417
13. Han JY, Park K, and Kim SW, et al (2012) First-SIGNAL: first-line single-agent iressa versus gemcitabine and cisplatin trial in never-smokers with adenocarcinoma of the lung J Clin Oncol 30(10) 1122–1128 https://doi.org/10.1200/JCO.2011.36.8456 PMID: 22370314
14. Rosell R, Carcerand E, and Gervais R, et al (2012) Erlotinib versus standard chemotherapy as first-line treatment for European patients with advanced EGFR mutation-positive non-small-cell lung cancer (EURTAC): a multicenter, open-label, randomized phase 3 trial Lancet Oncol 13(3) 239–246 https://doi.org/10.1016/S1470-2045(11)70393-X PMID: 22285168
15. Sequist LV, Yang JCH, and Yamamoto N, et al (2013) Phase III study of afatinib or cisplatin plus pemetrexed in patients with metastatic lung adenocarcinoma with EGFR mutations J Clin Oncol 31(27) 3327–3334 https://doi.org/10.1200/JCO.2012.44.2806 PMID: 23816960
16. Noronha V, Patil VM, and Joshi A, et al (2020) Gefitinib versus gefitinib plus pemetrexed and carboplatin chemotherapy in EGFR-mutated lung cancer J Clin Oncol 38(2) 124–136 https://doi.org/10.1200/JCO.19.01154
17. Soria JC, Ohe Y, and Vansteenkiste J, et al (2018) Osimertinib in untreated EGFR-mutated advanced non-small-cell lung cancer N Engl J Med 378(2) 113–125 https://doi.org/10.1056/NEJMoa1713137
18. Ramalingam SS, Vansteenkiste J, and Planchard D, et al (2020) Overall survival with osimertinib in untreated, EGFR-mutated advanced NSCLC N Engl J Med 382(1) 41–50 https://doi.org/10.1056/NEJMoa1913662
19. Planchard D, Janne PA, and Cheng Y, et al (2023) Osimertinib with or without chemotherapy in EGFR-mutated advanced NSCLC N Engl J Med 389(21) 1935–1948 https://doi.org/10.1056/NEJMoa2306434 PMID: 37937763
20. Cho BC, Felip E, and Hayashi H, et al (2022) MARIPOSA: phase 3 study of first-line amivantamab + Lazertinib versus osimertinib in EGFR-mutant non-small-cell lung cancer Future Oncol 18(6) 639–647 https://doi.org/10.2217/fon-2021-0923
21. Kobayashi S, Boggon TJ, and Dayaram T, et al (2005) EGFR mutation and resistance of non-small-cell lung cancer to gefitinib N Engl J Med 352(8) 786–792 https://doi.org/10.1056/NEJMoa044238 PMID: 15728811
22. Sequist LV, Waltman BA, and Dias-Santagata D, et al (2011) Genotypic and histological evolution of lung cancers acquiring resistance to EGFR inhibitors Sci Transl Med 3(75) 75ra26 https://doi.org/10.1126/scitranslmed.3002003 PMID: 21430269 PMCID: 3132801
23. Yu HA, Arcila ME, and Rekhtman N, et al (2013) Analysis of tumor specimens at the time of acquired resistance to EGFR-TKI therapy in 155 patients with EGFR-mutant lung cancers Clin Cancer Res 19(8) 2240–2247 https://doi.org/10.1158/1078-0432.CCR-12-2246 PMID: 23470965 PMCID: 3630270
24. Campo M, Gerber D, and Gainor JF, et al (2016) Acquired resistance to first-line Afatinib and the challenges of prearranged progression biopsies J Thorac Oncol 11(11) 2022–2026 https://doi.org/10.1016/j.jtho.2016.06.032 PMID: 27553514
25. Soria JC, Wu YL, and Nakagawa K, et al (2015) Gefitinib plus chemotherapy versus placebo plus chemotherapy in EGFR-mutation-positive non-small-cell lung cancer after progression on first-line gefitinib (IMPRESS): a phase 3 randomised trial Lancet Oncol 16(8) 990–998 https://doi.org/10.1016/S1470-2045(15)00121-7 PMID: 26159065
26. Remon J, Besse B, and Aix SP, et al (2023) Osimertinib treatment based on plasma T790M monitoring in patients with EGFR-mutant non-small-cell lung cancer (NSCLC): eORTC lung cancer group 1613 APPLE phase II randomized clinical trial Ann Oncol 34(5) 468–476 https://doi.org/10.1016/j.annonc.2023.02.012 PMID: 36863484
27. Tabara K, Kanda R, and Sonoda K, et al (2012) Loss of activating EGFR mutant gene contributes to acquired resistance to EGFR tyrosine kinase inhibitors in lung cancer cells PLoS One 7(7) e41017 https://doi.org/10.1371/journal.pone.0041017 PMID: 22815900 PMCID: 3398867
28. Ueda M, Namba M, and Tokumo K, et al (2022) Conversion from positive to negative EGFR mutation due to clonal selection during long-term treatment with epidermal growth factor receptor-tyrosine kinase inhibitors: a case report Case Rep Oncol 14(3) 1447–1453 https://doi.org/10.1159/000518246
29. Marcoux N, Gettinger SN, and O’Kane G, et al (2019) EGFR-mutant adenocarcinomas that transform to small-cell lung cancer and other neuroendocrine carcinomas: clinical outcomes J Clin Oncol 37(4) 278–285 https://doi.org/10.1200/JCO.18.01585
30. Lee CK, Robichaux JP, and Janne PA, et al (2023) Acquired mechanisms of resistance to first-line (1L) osimertinib with or without platinum-based chemotherapy (CT) in EGFR-mutated (EGFRm) advanced NSCLC: preliminary data from FLAURA2 Ann Oncol 34(S4) https://doi.org/10.1016/j.annonc.2023.10.593
31. Vokes NI, Chambers E, and Nguyen T, et al (2022) Concurrent TP53 mutations facilitate resistance evolution in EGFR-mutant lung adenocarcinoma J Thorac Oncol 17(6) 779–792 https://doi.org/10.1016/j.jtho.2022.02.011 PMID: 35331964 PMCID: 10478031
32. Offin M, Somwar R, and Rekhtman N, et al (2018) Acquired ALK and RET gene fusions as mechanisms of resistance to osimertinib in EGFR -mutant lung cancers JCO Precis Oncol 2 1–12 https://doi.org/10.1200/PO.18.00126
33. Papadimitrakopoulou VA, Wu YL, and Han JY, et al (2018) LBA51 analysis of resistance mechanisms to osimertinib in patients with EGFR T790M advanced NSCLC from the AURA3 study Ann Oncol 29 https://doi.org/10.1093/annonc/mdy424.064
34. Ramalingam SS, Cheng Y, and Zhou C, et al (2018) LBA50 mechanisms of acquired resistance to first-line osimertinib: Preliminary data from the phase III FLAURA study Ann Oncol 29
35. Lambros L, Guibourg B, and Uguen A (2018) ROS1-rearranged non-small cell lung cancers with concomitant oncogenic driver alterations: about some rare therapeutic dilemmas Clin Lung Cancer 19(1) e73–e74 https://doi.org/10.1016/j.cllc.2017.08.005
Supplementary data
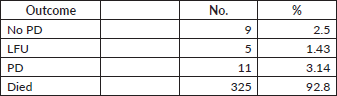
As per May 2024, the updated data shows the following outcomes – as charted in Table S1.
Table S1. Outcomes.
PFA analysis
Using Kaplan meir analysis and log rank test analysis to calculate the hazard ratio using progression, death or date of last follow-up as the event with the event occurring first being taken. The PFS for the entire cohort is 15.3 months (95% CI, 13.9 to 16.7 months). The addition of pemetrexed and carboplatin to geftinib improved the PFS from 12 months (95% CI, 10.9 to 13.4 months) to 19 months (95% CI, 16.4 to 21.4 months) with HR for death or progression being 0.52 (95% CI, 0.39 to 0.66; p < 0.001). The differences seen by the Kaplan meir analysis were noted to be significant by the, log rank test. The PFS among various groups was charted as follows in the Table S2.
Table S2. PFS among various groups.
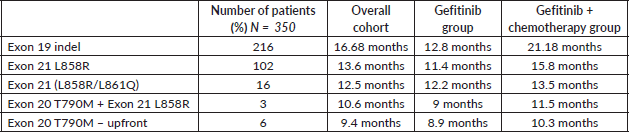
Site of progression
The radiological evaluation was done in all the patients suspected of clinical progression and in some patients as part of regular monitoring. In three patients, cerebro-spinal fluid (CSF) analysis revealed disease progression. The sites of progression were noted as follows in the evaluation – in Table S3.
Table S3. Sites of progression.
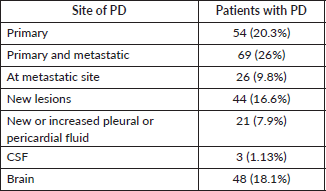
Site of biopsy
Based on the site of progression and feasibility of biopsy, biopsy or cell block was obtained. The sites of biopsy are charted in Table S4.
Table S4. Site of biopsy.
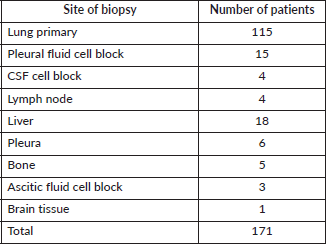
Complications of biopsy
Out of the 115 patients who had lung biopsy, 7 had minimal pneumothorax, 6 needed interventions due to pneumothorax, 1 patient had dizziness and bradycardia and 1 had severe post procedural pain. Of the others only one patient of liver biopsy had significant post procedural pain. The details are tabulated in Table S5.
Table S5. Complications of biopsy.
Molecular analysis of the 206 patients available for analysis
In the molecular analysis, T790M was noted as the predominant molecular alteration noted accounting for 61 patients being 29.6% of the cases with resistance Table S6.
Table S6. Molecular analysis of the 116 patients with new molecular alterations.
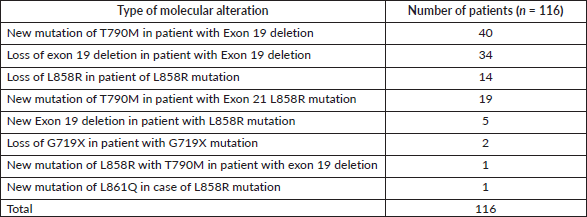
In rest of the patients- N -90
Table S7. Molecular analysis of the 90 patients with retained molecular alterations.

Additional mutation noted on NGS in 10 patients out of 17 in which NGS was performed Table S8.
Table S8. Additional molecular analysis noted on NGS.

Overlapping resistance patterns noted
- 1 case of SCLC with loss of exon 19 deletion
- 1 case of SCLC with new mutation of S768I with prior L858R retained
- 2 cases of SCLC with TP53 mutations
- 1 case of Neuroendocrine carcinoma transformation with T790M muatation
- 1 case of Adenocarcinoma to squamous cell carcinoma transformation along with T790M mutation
- 1 case of loss of exon 19 deletion with ALK mutation
- 1 case of loss of exon 19 deletion with ROS fusion
- 1 case of loss of L858R mutation with PIK3CA amplification
- 2 cases of loss of exon 19 deletion with gain of TP53 mutation
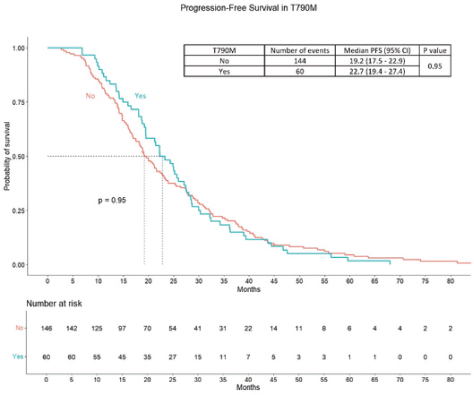
Figure S1. PFS in patients of T90M positive compared to other patients.
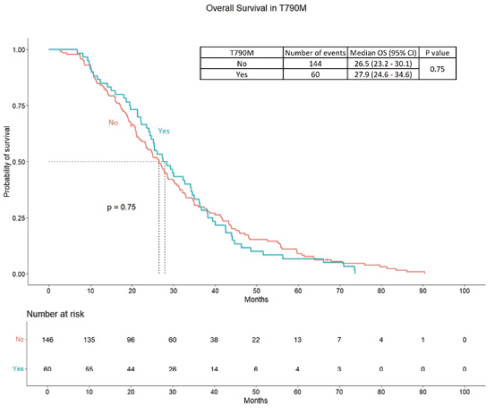
Figure S2. OS in patients of T90M positive compared to other patients.
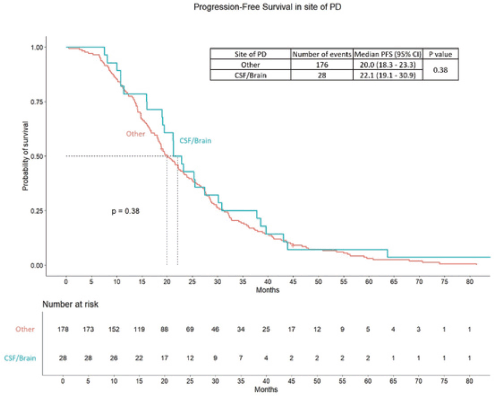
Figure S3. PFS difference between patients progressed in brain or CSF versus other sites of PD.
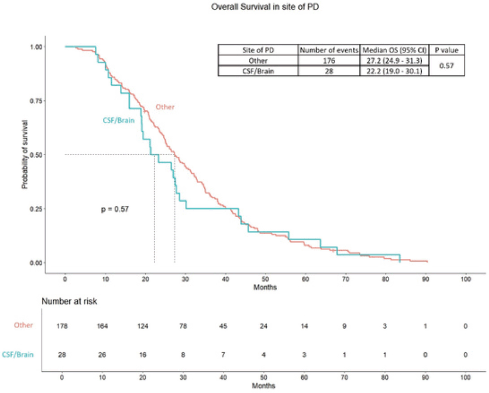
Figure S4. OS difference between patients progressed in the brain or CSF versus other sites of PD.