Low-energy ionizing radiation and carcinogenesis: a call for optimized clinical stewardship in the computed tomography era
Satyajeet Ratha, Samarth Ozab, Sweta Sonic and Sneha Dhillon
Department of Radiation Oncology, All India Institute of Medical Sciences, Rajkot 360060, India
a https://orcid.org/0000-0003-3905-4922
a https://orcid.org/0009-0009-5727-2692
a https://orcid.org/0000-00001-8349-1957
Abstract
Computed tomography (CT) scans are widely used diagnostics. Despite their diagnostic advantages, their increasing reliance necessitates critical examination of projected lifetime cancer risks due to radiation exposure, a stochastic effect linearly correlated with dose. CT-associated cancers are estimated to increase significantly. The paediatric population is disproportionately vulnerable, facing higher risks even from lower doses. Current risk models may underestimate cancer incidence due to Dose and Dose Rate Effectiveness Factor complexities. High-risk scans such as whole-body CT, abdominal CT and brain CT, significantly increase exposure. A critical re-evaluation of guidelines to prioritise the risk-benefit ratio for CT use. Future diagnostic and screening strategies must prioritize innovation in alternative non-ionizing modalities like magnetic resonance imaging, ultrasonography and thermography with integration of Artificial Intelligence. ‘As Low As Reasonably Achievable’ principle is essential, aiming to limit ionizing radiation doses to near zero to safeguard public health.
Keywords: cancer epidemiology, computed tomography, radiation protection
Correspondence to: Samarth Oza
Email: samarthoza8@gmail.com
Published: 31/03/2026
Received: 24/07/2025
Publication costs for this article were supported by ecancer (UK Charity number 1176307).
Copyright: © the authors; licensee ecancermedicalscience. This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
First clinical computed tomography (CT) Scan was used in 1971, and since then it has became a popular diagnostic tool due to its superiority not only in sensitivity and specificity but also cost effectiveness, lesser scan time and universal availability [1, 2]. Compared to its peers, e.g., ultrasonography (USG) and magnetic resonance imaging (MRI), which were also introduced in clinical use in 1970s and 1980s, respectively, CT Scan is admired due to its universal diagnostic capabilities and financial suitability. An estimated 93 million CT scans, nearly 40 million MRIs, and more than 100 million ultrasonography examinations were performed annually in the USA as per 2023 data. [3, 4]. The stochastic effect of radiation is a well-established principle, and it demonstrates a linear correlation between radiation doses and increased cancer risk [5]. Equation for cancer risk:
R cancer = Effective dose (Sv) × Risk factor (5.5 × 10−2) (F) (ICRP)
Radiation exposure increases the risk of cardiovascular diseases and benign tumours, although current data are inadequate for risk quantification (BEIR VII) [6].
The increasing reliance on CT imaging, while a diagnostic triumph, necessitates a critical examination of its projected lifetime cancer risks. It is estimated that over 100,000 new cancer cases in the United States each year may be attributable to exposure from CT imaging, representing approximately 5% of all newly diagnosed cancers [4]. Notably, the paediatric population, especially those under 1 year of age, faces a disproportionately higher projected cancer risk, even from lower radiation exposure [6]. An Australian epidemiological study on paediatric population (0–19 age) estimated approximately 24% increased cancer incidence per CT scan [7]. This heightened vulnerability in children is attributed to their longer life expectancy, allowing more time for radiation-induced cancers to manifest and their greater cellular radio sensitivity [8]. A retrospective hospital study on surgical patients revealed that roughly 50% of CT scans were unremarkable [9], highlighting potential overuse and unnecessary radiation exposure.
The biological effects of ionizing radiation were mainly studied on The Japanese atomic bomb survivors and scientific committee BEIR VII reported that radiation induced carcinogenesis follows ‘Linear-No-Threshold’ (LNT) risk model meaning there will be ‘some’ risk even at lower doses of exposure. BEIR VII reported life time cancer risk of one in 100 individuals for single exposure of 100 mSv. This risk decreased proportionately as one in 1,000 individuals for single exposure of 10 mSv [10]. Current reports estimate lifetime cancer risk from CT scan based on standard BEIR VII and Radiation Risk Assessment Tool (RadRAT). RadRAT calculator was developed by National Cancer Institute which provides lifetime cancer risk and organ-specific risk estimates. As radiosensitivity and repair capacity of various organs are different, similar low dose exposure may exhibit different effects in organs. Schneider et al [11] proposed organ equivalent dose as LNT model does not effectively estimate cancer risk of specific organs except colon, cervix and skin. They reported excess relative risk of 1, 0.85 and 0.48 events per Gray (Gy) for female breast, colon and lung, respectively, for exposure at age of 30 and attained age of 70 years [11]. On a practical note, one individual may be exposed to different types of low-energy modalities (X Rays, Fluoroscopy, CT, Positron Emission Tomography and so on) at different intervals and to different body parts. Cumulative exposure doses may vary depending on individual bases. Ionizing Radiation Reporting System should be established, which collects site specific and organ-specific data for individuals and from all low-energy ionizing radiation types. This will generate more accurate data for more precise estimation. Sodickson et al [12] studied on cumulative CT scan doses in which 15% patients received ≥100 mSv and 4% patients received 250–1,375 mSv of cumulative exposure. A study showed additional cancer risk of 1 per 1,000 patients for 10 mSv exposure at age of 30 for females and 20 for males. For females below the age of 30 and males below the age of 20 excess cancer risk follows exponential trajectory with the highest risk reaching upto ~4.5 per 1,000 females and ~2.5 per 1,000 males [12]. Similarly, repeat CT scans done at intervals of 6–12 weeks can result in elevated levels of DNA repair foci which can elevate risk of malignancy [13]. In addition to diagnostic scans, imaging done to verify setup during radiotherapy treatment delivery also adds to the cumulative lifetime radiation dose. Specifically in Image Guided Radiation Therapy (IGRT) includes low energy cone beam CT (kvCBCT) scans for accurate matching and dose delivery. Kim et al [14] reported association of kvCBCT imaging doses and secondary cancer risk. They found lifetime attributed risk for in-field organs is in range of ~100–400 per 10,000 and for out-of-field organs it is ~2–4 per 10,000 for the pelvic region site (average in-field dose:16.7 cGy, average out-of-field dose: 0.67–0.02cGy) [14]. It shows IGRT imaging optimization is very important, specifically for paediatric age group.
Dose and Dose Rate Effectiveness Factor (DDREF) depends on two variables: Low Dose Effective Factor and Dose rate Effective Factor. Where low dose is defined as less than 100 mGy and low dose rate is defined as less than 1 mGy/min averaged over 1 hour. ICRP in 2007 accepted DDREF 2 as standard but various international bodies were not in agreement. BEIR VII and NAS in 2006 considered DDREF value as 1.5. Whereas WHO in 2013 reported use DDREF value 1 as standard. UNSCEAR was against the use of DDREF for calculation [15]. Impact on risk estimates is huge when DDREF values changes as estimates doubles when factor value one is used instead of two. Observed DDREF in nuclear worker epidemiological study done by Hoel et al [16] suggest DDREF 2.63. Nonlinear dose response curves at lower dose along with bystander effects makes it more complicated to understand effects of radiation at cellular level which may continue for many years or even decades [17]. Integration of epidemiological & radiobiological research is need of hour for stronger evidence [17]. Lower doses of radiation may cause non-cancerous effects such as inflammatory responses, which may be beneficial or harmful [18]. Recent advances in clinical radiation oncology e.g. FLASH radiotherapy also highlights very high dose rates effects are not comparable to routine conventional dose rate effects. To generate stronger and specific evidence epidemiological studies are essential.
Table 1. Ionizing radiation and approximate effective radiation dose [21].
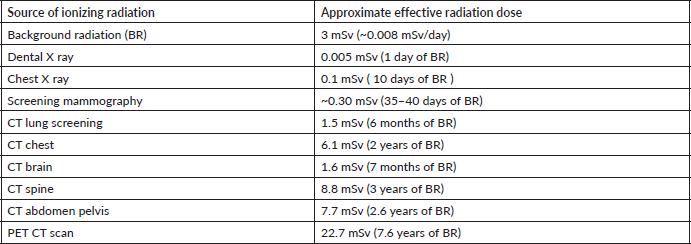
With increasing life expectancy due to advancement in healthcare, use of diagnostic imaging is set to rise further. We must critically re-evaluate the risk-benefit ratio for CT use, particularly in paediatric age and for whole-body, abdominal and brain CT scans in adult age, given their association with higher cancer risk (Table 1).
Adherence to the ‘As Low As Reasonably Achievable’ principle for radiation protection is paramount. Screening CT adds considerable numbers of scans; therefore, the use of alternative screening tools should be encouraged. With 42 million screening mammograms performed last year in the USA alone [19], there is a compelling concern that this widespread screening contributes to the rising global incidence of breast cancer observed since its introduction [20]. Future strategies must prioritize innovation in alternative screening and diagnostic techniques – such as MRI, USG and thermography (for breast) – integrating Artificial Intelligence/Machine Learning to achieve comparable sensitivity, specificity, cost-effectiveness and feasibility to CT.
Our ultimate goal should be to limit ionising radiation doses to as close to zero as possible, safeguarding public health in the long term.
The following CT Usage Recommendations may be followed to minimize exposure:
(a) Prefer alternative non-ionising imaging modalities, particularly for the paediatric population
(b) Consideration of cumulative dose accumulation factor before prescribing scan – that requires patient-based unique data for the previous scan and exposure details
(c) In cases where CT imaging cannot be avoided, radiation dose optimization should be achieved by utilizing the latest hardware improvements and AI/ML-based software techniques.
(d) Hospital-based monthly and annual review of number of scans and reason for scan, particularly for paediatric age group.
Conflicts of interest
The authors declares no conflicts of interest.
Funding
No funding was obtained or allocated for the conduct of this study.
Acknowledgments
None.
References
1. Beckmann EC (2006) CT scanning the early days Br J Radiol 79(937) 5–8 https://doi.org/10.1259/bjr/29444122 PMID: 16421398
2. Schulz RA, Stein JA, and Pelc NJ (2021) ‘How CT happened: the early development of medical computed tomography’ J Med Imag 8(05) https://doi.org/10.1117/1.JMI.8.5.052110
3. Shah A and Aran S (2023) A review of magnetic resonance (MR) safety: the essentials to patient safety Cureus 15(10) e47345 http://doi.org/10.7759/cureus.47345 PMID: 38021512 PMCID: 10657250
4. Smith-Bindman R, Chu PW, and Azman Firdaus H, et al (2025) Projected lifetime cancer risks from current computed tomography imaging JAMA Internal Med 185(6) 710 https://doi.org/10.1001/jamainternmed.2025.0505
5. Vetter R (2007) The 2007 recommendations of the ICRP Health Phys 95(3) 472–474 http:doi.org/10.1097/01.HP.0000324200.73903.5b
6. Little MP, Azizova TV, and Richardson DB, et al (2023) ‘Ionising radiation and cardiovascular disease: systematic review and meta-analysis’ BMJ 380 e072924 https://doi.org/10.1136/bmj-2022-072924
7. Mathews JD, Forsythe AV, and Brady Z, et al (2013) Cancer risk in 680 000 people exposed to computed tomography scans in childhood or adolescence: data linkage study of 11 million Australians BMJ 346(may21 1) f2360 https://doi.org/10.1136/bmj.f2360
8. Sadetzki S and Mandelzweig L (2009) Childhood exposure to external ionising radiation and solid cancer risk Br J Cancer 100(7) 1021–1025 https://doi.org/10.1038/sj.bjc.6604994 PMID: 19337255 PMCID: 2669984
9. Tanveer Y, Ahmed F, and Aslam A, et al (2022) Retrospective audit of CT scans performed at a hospital for surgical patients Ann Med Surg 78 https://doi.org/10.1016/j.amsu.2022.103788
10. National Research Council; Division on Earth and Life Studies; Board on Radiation Effects Research; Committee to Assess Health Risks from Exposure to Low Levels of Ionizing Radiation (2005) Health risks from exposure to low levels of ionizing radiation: BEIR VII phase 2. BEIR VII Phase 2 https://nap.nationalacademies.org/catalog/11340/health-risks-from-exposure-to-low-levels-of-ionizing-radiation
11. Schneider U, Sumila M, and Robotka J (2011) Site-specific dose-response relationships for cancer induction from the combined Japanese A-bomb and Hodgkin cohorts for doses relevant to radiotherapy Theor Biol Med Model 8(1) https://doi.org/10.1186/1742-4682-8-27 PMID: 21791103 PMCID: 3161945
12. Sodickson A, Baeyens PF, and Andriole KP, et al (2009) Recurrent CT, cumulative radiation exposure, and associated radiation-induced cancer risks from CT of adults Radiology 251(1) 175–184 https://doi.org/10.1148/radiol.2511081296 PMID: 19332852
13. Bogdanova NV, Jguburia N, and Ramachandran D, et al (2021) Persistent DNA double-strand breaks after repeated diagnostic CT scans in breast epithelial cells and lymphocytes Front Oncol 11 https://doi.org/10.3389/fonc.2021.634389 PMID: 33968734 PMCID: 8103218
14. Kim DW, Chung WK, and Yoon M (2013) Imaging doses and secondary cancer risk from kilovoltage cone-beam CT in radiation therapy Health Phys 104(5) 499–503 https://doi.org/10.1097/HP.0b013e318285c685 PMID: 23532078
15. ICRP [Internet]. Icrp.org. https://icrp.org Date accessed: 16/07/2025
16. Hoel DG (2018) Nuclear epidemiologic studies and the estimation of DREF Int J Radiat Biol 94(4) 307–314 https://doi.org/10.1080/09553002.2018.1437483 PMID: 29400635
17. Rühm W, Azizova TV, and Bouffler SD, et al (2016) Dose-rate effects in radiation biology and radiation protection [Internet] Ann ICRP 45(1_suppl) 262–279 https://doi.org/10.1177/0146645316629336
18. Lowe D, Roy L, and Tabocchini MA, et al (2022) Radiation dose rate effects: what is new and what is needed? Radiat Environ Biophys 61(4) 507–543 https://doi.org/10.1007/s00411-022-00996-0 PMID: 36241855 PMCID: 9630203
19. Health C for D and R (2024) MQSA National Statistics [Internet] (FDA) https://www.fda.gov/radiation-emitting-products/mammography-information-patients/mqsa-national-statistics
20. Ilic L, Haidinger G, and Simon J, et al (2022) Trends in female breast cancer incidence, mortality, and survival in Austria, with focus on age, stage, and birth cohorts (1983–2017) Scientific Rep 12(1) 12 http:doi.org/10.1038/s41598-022-10560-x
21. Radiology info (2021) Radiation Dose in X-Ray and CT Exams [Internet] (American College of Radiology) [https://www.radiologyinfo.org/en/info/safety-xray]