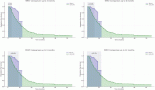
The use of small molecule inhibitors in acute myeloid leukaemia (AML) has become ubiquitous with the US Food and Drug Administration approval of multiple agents between 2017 and 2018. Despite the promise, some of these indications are based on early efficacy data (phase I/II), and single-arm studies, and have not been tested in randomised trials. Furthermore, there are important limitations in the evidence that exists in randomised trials. This perspective aims to summarise the data that formed the basis for approval of gilteritinib, glasdegib, ivosidenib, enasidenib and venetoclax. It also aims to shed a light on some of the limitations in the evidence. Clinicians should exercise caution when using drugs that largely have yet to show an improvement in survival over the standard of care in AML.