Disparities in oral cancer screening among Indian women aged 30–49: insights from a national survey
Navjot Kaur1a, Pritam Halder2b, Yuvaraj Krishnamoorthy3, Gursimer Jeet4, Garima Bhatt5, Sathish Rajaa6c, Priyanka Sudhir7, Rohit Sharma8,9, Sarthak Tandon10d, Manish Gupta1e
1Otorhinolaryngology and Head Neck Surgery, Dr B R Ambedkar State Institute of Medical Sciences (AIMS, Mohali), Sector 56 A, SAS Nagar, Punjab 160055, India
2Department of Community Medicine and School of Public Health, Postgraduate Institute of Medical Education and Research, Chandigarh 160012, India
3Evidence Synthesis Unit, Partnership for Research, Opportunities, Planning, Upskilling and Leadership (PROPUL) Evidence, Chennai 600099, India
4Vancouver, V6M1T5, Canada
5Department of Health Sciences, University of York, YO10 5DD York, UK
6Department of Community Medicine, ESIC Medical College & PGIMSR, K.K. Nagar, Chennai 600078, India
7Sacramento, CA, USA
8Al Muhaidb Dental Clinic, Jazan 82724, Saudi Arabia
9Department of Oral and Maxillofacial Surgery, DMIHER (DU) Wardha 442001, Maharashtra, India
10Department of Radiation Oncology, Rajiv Gandhi Cancer Institute and Research Centre, Delhi 110085, India
ahttps://orcid.org/0000-0002-5563-724X
bhttps://orcid.org/0009-0008-2407-6807
chttps://orcid.org/0000-0002-2319-0391
dhttps://orcid.org/0000-0002-2319-0391
ehttps://orcid.org/0000-0001-7784-308X
Abstract
Introduction: Oral cancer estimates are concerning in India, with inequalities in accessing screening services, especially in rural areas. Socioeconomic characteristics contribute to disparities in screening coverage. The current study estimates the coverage of self-reported screening, spatial patterns, differences in screening rates in urban and rural areas and determinants of screening among Indian women.
Methodology: We analysed data from 348,882 women (30–49 years) participating in India’s fifth wave of the National Family Health Survey (NFHS-5). Self-reported oral cancer screening weighted coverage was estimated and compared per socio-demographic characteristics. Global and local spatial autocorrelation methods were applied to understand the spatial distribution of screening coverage, which was then depicted using choropleth maps. The differences in urban-rural screening were decomposed and determinants of screening were identified using the multivariable binary logistic regression. Analysis was done using Stata v17.0.
Results: Overall, at the national level, self-reported screening coverage was 0.87%, with higher rates in urban areas (1.08%) compared to rural areas (0.77%). Screening uptake increased with age, socioeconomic status and education. Scheduled Tribes and the poorest quintile had the lowest rates. 348,882 participants were included in the final analysis after all exclusions. The uptake of oral cancer screening increased with an increase in wealth Index (Middle: adjusted odds ratio: 1.35; 95% CI: 1.07–1.70), Richer (1.43; 1.12–1.84), Richest (1.60; 1.20–2.13) and in obese women (1.28; 1.02–1.63). Meanwhile, women who belonged to the Muslim religion (0.68; 95% CI: 0.56–0.84), scheduled tribes (0.70; 95% CI: 0.53–0.84) and those who were illiterate (0.66; 0.51–0.85) had lower odds of screening uptake. Women from South Indian states (9.58; 95% CI: 7.60–12.07), West Indian states (3.81; 95% CI: 2.88–5.04), Central India (2.48; 95% CI: 1.95–3.14) and North-east Indian states (1.65; 95% CI: 1.20–2.27) had higher odds of oral cancer screening uptake compared to North Indian states. The urban-rural gap was 57.76% due to factor distribution and 42.24% due to differences in factor effects. Religion, caste, education and media exposure all significantly contributed to the gap.
Conclusion: Screening uptake varied according to socio-economic status and region of the country. Significant disparities in oral cancer screening exist among urban and rural women, driven by socioeconomic factors. Enhancing healthcare access, education and media outreach in rural areas is essential to improving screening rates and reducing disparities.
Keywords: inequalities in health, disparities, inaccessibility, oral cancer, national survey, women’s health
Correspondence to: Navjot Kaur and Gursimer Jeet. Email: navjot517@gmail.com and dr.gursimerjeet@gmail.com
Published: 18/03/2026
Received: 09/09/2025
Publication costs for this article were supported by ecancer (UK Charity number 1176307).
Copyright: © the authors; licensee ecancermedicalscience. This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
With increasing life expectancy and changes in lifestyle patterns, the global burden of disease has shifted from infectious to chronic non-communicable diseases (NCDs), among which cancers have become increasingly prominent [1]. This epidemiological transition poses substantial challenges for health systems worldwide, with cancers contributing to nearly 19.8 million new cases and 9.74 million deaths annually and resulting in a loss of an estimated 250 million disability-adjusted life years [2]. Oral cancers affecting the mucosal surfaces of the lips, tongue, floor of the mouth and buccal mucosa have gained particular salience and rank among the top 20 most common cancers globally [3]. According to GLOBOCAN 2022, an estimated 377,713 new cases and 177,757 deaths are attributed to oral cancers globally, with South and Southeast Asia bearing the highest burden [4]. These estimates are overwhelming, particularly in resource-constrained countries. India alone contributes nearly one-third of the global oral cancer burden, accounting for 13%–16% of all cancers nationally [5, 6]. Most recent estimates from Indian population-based cancer registry data indicate that 63,671 women reported new oral cavity and pharyngeal cancer cases in 2024, corresponding to a crude incidence rate of 9.2 per 100,000 and a lifetime risk of about 1%, with mouth and tongue cancers forming the major subsites; additionally, these cancers contributed to approximately 37,418 deaths among women in 2024, reflecting a crude mortality rate of 5.4 per 100,000 [7].
Socioeconomic disparities play a critical role in the epidemiology of oral cancers in India, with the disease disproportionately affecting disadvantaged populations. Prominent risk factors such as widespread use of smokeless tobacco products like mishri, gutkha and tobacco paan, along with human papillomavirus infections, are strongly implicated in the high incidence of oral precancerous lesions and cancers [8]. Additionally, gender-specific risk behaviours and exposures, including increasing tobacco use among women and poor oral hygiene, are contributing to the rising burden among females, particularly in rural areas [9]. Presentation in late stages and inadequate access to diagnostic and treatment facilities, especially among females, lead to poor survival rates [10]. Early detection remains pivotal, as 5-year survival rates exceed 80% when diagnosed early, but drop to 20%–30% for late-stage diagnoses [11].
Recognising these challenges, the World Health Organisation and the Indian Council of Medical Research recommend integrating visual oral cancer screening into primary healthcare through both population-based and opportunistic approaches [12]. Randomised community-based trials in India have demonstrated that trained frontline health workers can effectively perform visual oral examinations, detect premalignant lesions and significantly reduce mortality from oral cancers [13]. Despite this evidence, national screening uptake remains extremely low. The fifth round of the National Family Health Survey (2019–21) report indicates that only about 1.2% of women aged 15–49 years have ever undergone oral cancer screening, with widespread urban-rural geographic disparities [14]. This is concerning, given that tobacco use and related risk factors remain more prevalent among rural women, as documented by recent rounds of the Global Adult Tobacco Survey [15–17]. Additional barriers such as lower health literacy, sociocultural norms, gendered restrictions on mobility and decision-making and disparities in primary healthcare access further impede screening uptake in rural settings [12, 18].
Although oral cancer incidence is higher among men, studying women separately is essential because they face distinct structural and gender-linked barriers that disproportionately limit screening uptake. Women often have lower autonomy, restricted mobility and reduced decision-making power regarding their health, contributing to delayed care-seeking and poorer outcomes. Moreover, India’s population-based NCD screening platforms, delivered largely through frontline workers and reproductive-health-aligned community systems, interact more consistently with women, making them an operational priority group for early detection. A female-focused analysis, therefore, facilitates a clearer understanding of gender-specific inequities and supports the development of tailored, equity-oriented interventions [19, 20]. Given these concerns, understanding the socio-demographic and geographic determinants of oral cancer screening uptake among women is essential. Women aged 30–49 years represent the priority population for oral cancer screening under India’s National Programme for Prevention and Control of NCDs, and fifth wave of the National Family Health Survey (NFHS-5) provides complete and nationally representative data for this age group. Therefore, the present study was done to examine the coverage of oral cancer screening in urban and rural areas stratified by socio-demographic characteristics, determine the spatial patterns and identify any regional variations, ascertain the factors contributing to urban-rural disparities and those influencing the likelihood of oral cancer screening separately among women aged 30–49 years, residing in urban, rural and overall Indian settings.
Methodology
Study design and data source
We re-analysed a dataset from the NFHS-5, which is freely available in the public domain. The NFHS, India’s version of the Demographic Health Survey, began in 1992–93, providing nationally representative, de-identified and state- and gender-disaggregated data on maternal, child and later men’s health for policy analysis [14, 21, 22].
Study population
The NFHS prioritises maternal and child health, sampling 636,699 households with 724,115 women (15–49 years old) and 101,839 men (15–54 years old). Using multi-phase stratified cluster sampling, Census Enumeration Blocks and villages served as Primary Sampling Units, selected via the Probability Proportional to Size sampling technique [14]. The data were recorded electronically in local languages using different study tools. Specifically, the Women’s tool inquired about a broad range of subjects, including inquiries about screening for breast, cervical and oral cancer [14]. The Biomarker tool for women included measurements of height (in cm), weight (in kg), waist and hip circumference (in cm), haemoglobin (mg/dL), blood pressure (mm of hg) and random blood sugar measurements (mg/dL).
Study sample
Of the 724,115 women who participated in the NFHS-5, we excluded women <30 years (
Study variables
Self-reported oral cancer screening was our main dependent variable. The women were enquired using a direct question, ‘Have you
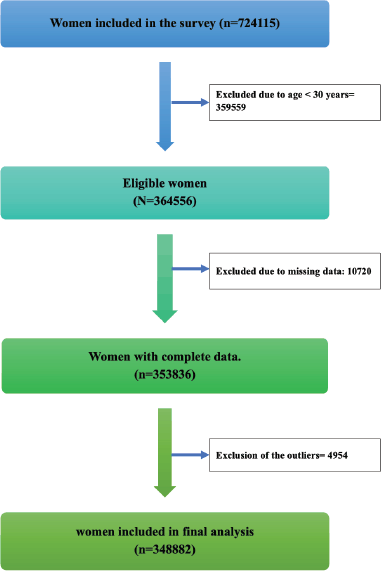
Figure 1. Sample selection process used in the study.
Data analysis
The analysis plan is based on the author’s previously published paper assessing disparities in cervical cancer screening among the women of reproductive age group of India [24]. We conducted a bivariate analysis using STATA v17.0 to calculate the coverage of self-reported oral cancer screening across different independent variables, reporting weighted proportions and their 95% confidence intervals. The associations were tested using a Chi-squared test. Both global and local spatial autocorrelation methods were applied to understand the spatial distribution of screening coverage. Global analysis using Moran’s I, based on Queen’s first-order matrix, assessed the overall spatial pattern – whether coverage was clustered, dispersed or randomly distributed using GeoDa software version 1.14. A significant pseudo-
Results
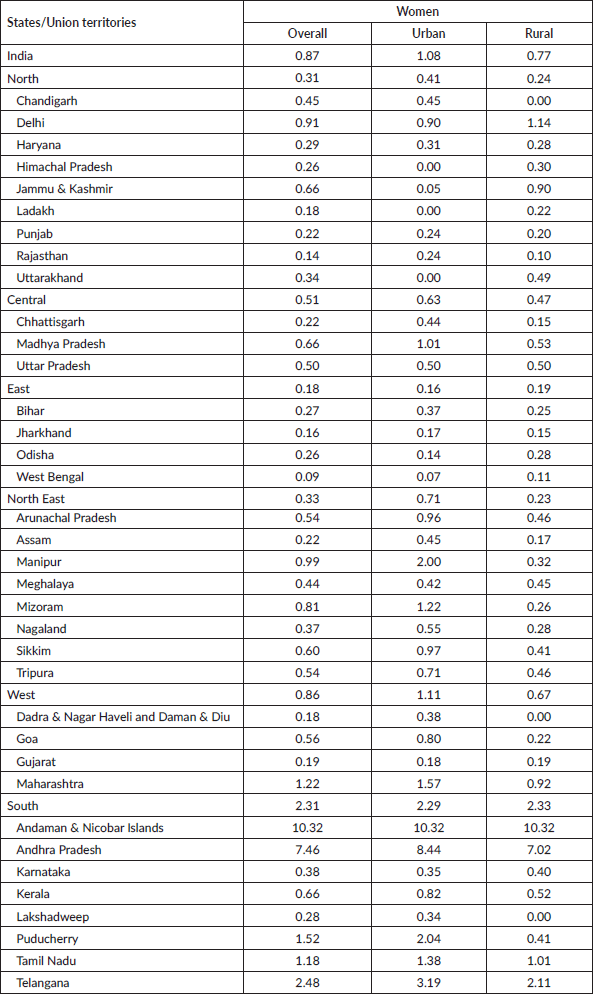
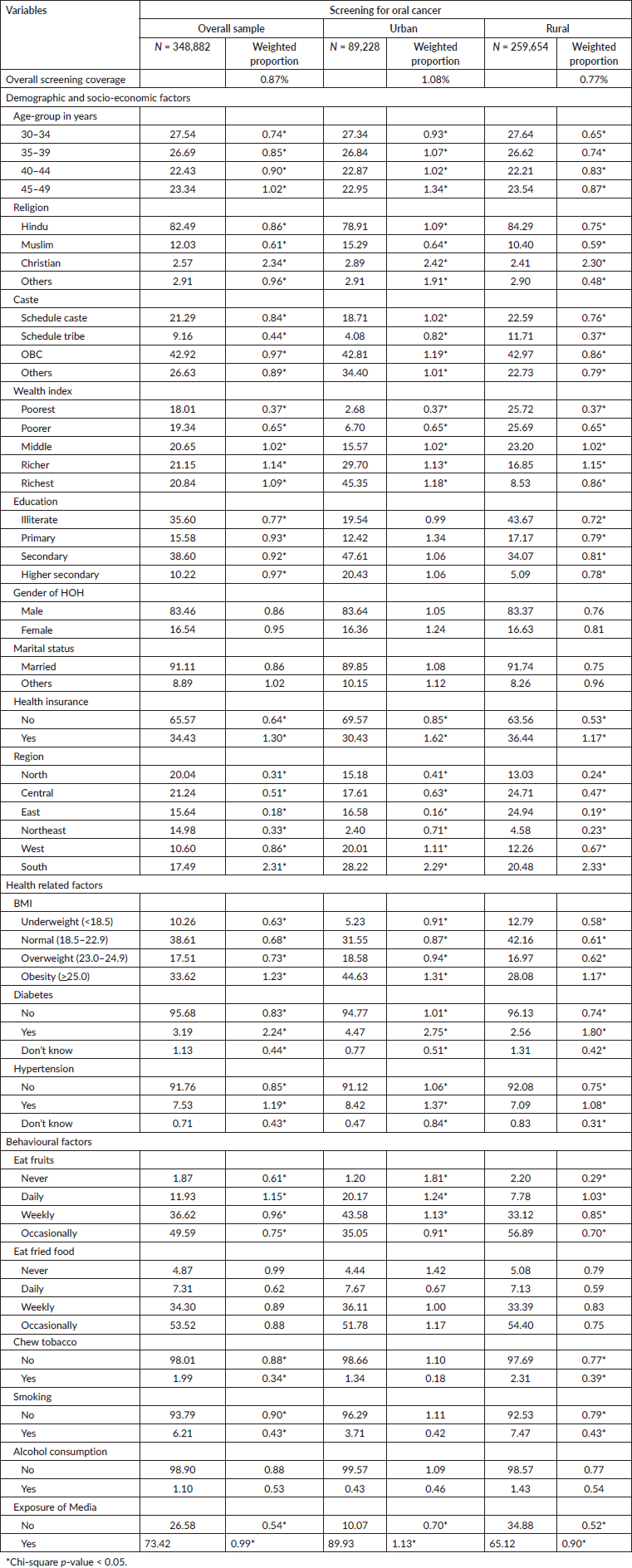
Of the 348,882 women analysed, the national coverage of self-reported screening for oral cancers was 0.87% (95% CI: 0.84–0.90). The rural areas (0.77%; 95% CI: 0.74–0.80) exhibited lower coverage than urban areas (1.08%; 95% CI: 1.04–1.12). Table 1 provides an elaboration of the coverage proportions by socio-demographic characteristics. The screening rates increase with age and peak in the 45- to 49-year-old age group. Urban women in this age group (1.34%), Christians (2.34% overall) and the richest urban quintile (1.18%) had a higher screening rate. Scheduled Tribes (0.44% overall) and the poorest quintile had the lowest screening rates (0.37% in urban and rural areas). Education levels were positively associated with screening rates. Female-headed households had higher screening rates, particularly in urban areas (1.24%). Married women also had higher screening rates, with urban areas showing a rate of 1.08% compared to 0.75% in rural areas. Health insurance significantly increased screening rates, especially in urban areas (1.62% versus 1.17% in rural areas). Women living with obesity (High BMI) had the highest screening rates (1.23%), with urban obese women having a rate of 1.31% compared to 1.17% in rural areas. Women with diabetes and hypertension also had higher screening rates, with urban women showing higher rates (2.75% and 1.37%, respectively) compared to rural women (1.80% and 1.08%, respectively). Behavioural factors such as daily fruit consumption were associated with higher screening rates, especially in urban areas (1.24% compared to 1.03% in rural areas). Tobacco users had lower screening rates, particularly in rural areas (0.39%). Smokers had lower screening rates overall, with rural smokers having a rate of 0.43%. Alcohol consumers had lower screening rates, with rural consumers showing a rate of 0.54%. Women exposed to media had significantly higher screening rates, particularly in urban areas (1.13% compared to 0.90% in rural areas) (Supplementary Table S1). Regional differences were evident, with Southern India having the highest screening rates (2.31% overall) and urban areas showing slightly lower rates (2.29%) compared to rural areas (2.33%). The Northeast region had a low overall screening rate (0.33%), with urban areas exhibiting a higher rate (0.71%) compared to rural areas (0.23%). Overall, West Bengal had the lowest coverage (0.09%). The lowest coverage in the urban and rural areas was revealed in Uttarakhand and Lakshadweep, respectively. Andaman and Nicobar Islands had the highest coverage (10.32% each) in urban and rural areas (Supplementary Table S2). There were spatial disparities in coverage as confirmed by Moran’s I statistic (0.667) (Figure 2). Higher coverage proportions, also known as hotspots, were spatially clustered in states like Kerala, Punjab and Jammu & Kashmir. On the contrary, low coverage or cold spots depicted clustering in states from the Central, Eastern and Northeastern regions (Figure 3).
Table 1. Self-reported oral cancer screening by women who participated in the fifth National Family Health Survey, India (2019–21).
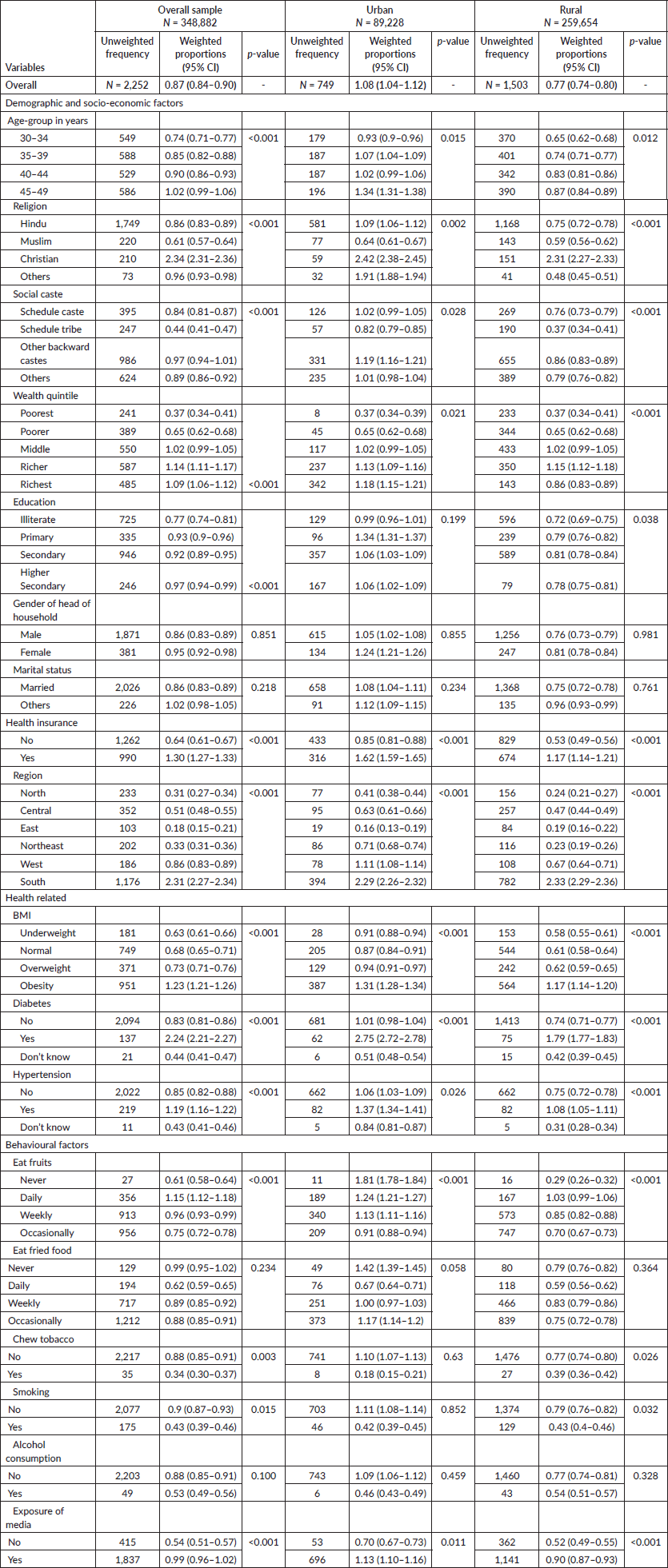
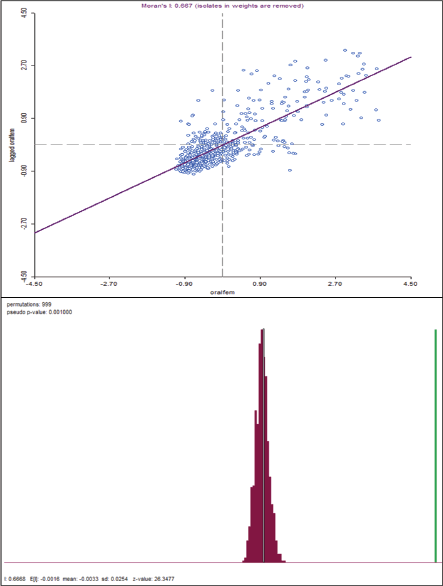
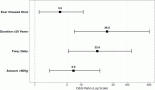
Figure 2. Moran’s I scatter plot showing spatial autocorrelation of oral cancer screening coverage among Indian women aged 30–49 years, along with permutation-based significance testing (999 permutations).
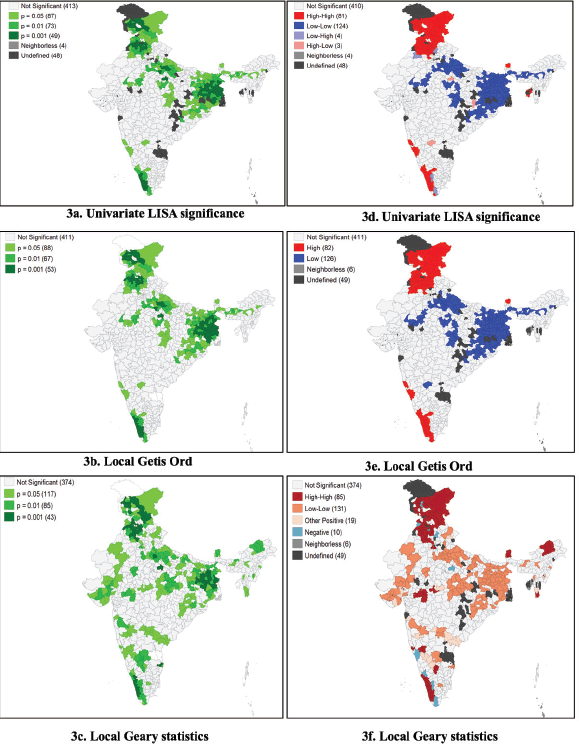
Figure 3. Spatial clustering of the oral cancer screening coverage (3a–c) and geographical clustering of hotspots and cold spots (3d–f).
Table 2 depicts the results from the decomposition analysis conducted to estimate the contributions of various independent variables to the urban-rural differences in the screening coverage. The overall urban-rural difference was statistically significant (
Table 2. Factors contributing to differences in urban-rural self-reported screening coverage among women participants of NFHS-5.
From the urban perspective, higher secondary education and health insurance reduced the gap by 9.17% and 5.91%, respectively. Among health-related factors, diabetes significantly contributed to widening the gap from the urban perspective by 3.57%. Among behavioural factors, daily consumption of fruits contributed to reducing the gap. Occasional fruit consumption widened the gap from the urban perspective and diminished it from the rural perspective. Tobacco positively contributed from the urban perspective by 2.22% and negatively from the rural perspective by −5.99%. Media exposure contributed to widening the gap from urban and rural perspectives by 3.87% and 58.62%, respectively. The South region had the most considerable positive contribution to the urban-rural gap, with a 33.73% increase from the urban perspective. The Eastern and Western regions made positive contributions, while the Central and Northeastern regions made negative contributions to the gap from an urban perspective.
Figure 4 illustrates the socio-demographic determinants of oral cancer screening among Indian women. The likelihood of oral cancer screening coverage increased with the wealth index from middle to richest (Middle wealth index, aOR 1.35 (95% Confidence Interval: 1.07–1.70), Richer (1.43; 95% CI: 1.12–1.84), Richest (1.60; 95% CI: 1.20–2.13)) compared to poorest. Illiterate women underwent less screening uptake for oral cancer as compared with those who had higher secondary educational status (0.66; 95% CI: 0.51–0.85). Christian women had higher odds, whereas Muslim women had lower odds for uptake of oral cancer screening compared to Hindu women (Christian: 1.45; 95% CI: 1.11–1.90, Muslim: 0.68; 95% CI: 0.56–0.84). Women who belonged to the Scheduled tribes had lower odds for screening uptake (0.70; 95% CI: 0.53–0.84). Obese women had higher uptake for screening compared with underweight women (1.28; 95% CI: 1.02–1.63). Women living in Central, North-east, West and Southern India had higher odds of screening uptake compared with North India (Central: 2.48; 95% CI: 1.95–3.14, North-east:1.65; 95% CI: 1.20–2.27, West: 3.81; 95% CI: 2.88–5.04, South: 9.58; 95% CI: 7.60–12.07).
Discussion
This study reveals disparities in self-reported oral cancer screening among Indian women aged 30–49 years, highlighting how structural and social health determinants shape access to preventive services. Using a nationally representative dataset and a PROGRESS-Plus informed lens, we found that factors such as place of residence, caster, education and socioeconomic status significantly shape screening behaviours, underscoring persistent inequities in India’s health system. The extremely low national average and persistent urban-rural divide in screening uptake reflect deeply entrenched barriers to preventive care services.
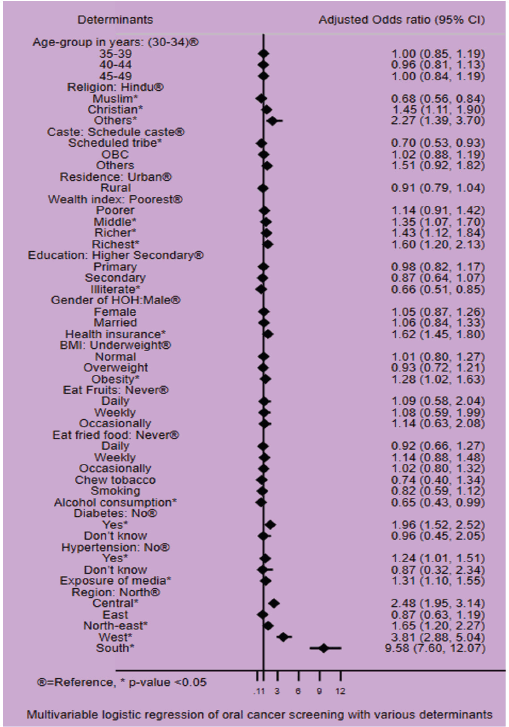
Figure 4. Determinants of oral cancer screening among Indian women participants (30–49 years) from the NFHS-5.
It is also important to interpret the low screening uptake in the context of the COVID-19 pandemic, as NFHS-5 fieldwork overlapped with multiple phases of nationwide lockdowns from March 2020 to April 2021 [32]. During this period, routine NCD services were widely disrupted due to mobility restrictions, repurposing of health workers for COVID duties, closure of outpatient services and fear of infection [33]. National reports indicate major declines in preventive and early-detection services during this period, suggesting that the NFHS-5 estimates reflect both true low coverage and temporary service interruptions.
Place of residence, a foundational PROGRESS variable, emerged as a critical driver of inequity. Rural women- particularly from marginalised communities- face limited availability of services, weaker health system infrastructure and sociocultural constraints that restrict access [34]. Despite the presence of recognised risk factors such as tobacco and alcohol use, overall screening uptake among Indian women remains extremely low, indicating that structural and gender-linked barriers outweigh individual risk profiles in shaping screening behavior [19, 20, 35]. Indian women commonly prioritise household responsibilities, childcare and family wellbeing over their own health needs, a pattern documented across chronic diseases and women’s cancers [36–38]. Studies consistently show that women delay or avoid preventive care due to limited autonomy, time constraints and dependence on family members for travel [39]. These gender-linked constraints likely contribute substantially to low oral cancer screening uptake.
Caste-based disparities, particularly for STs, persist as a marker of historical marginalisation. Women from tribal communities had the lowest screening rates (0.44%) with significantly lower odds of screening services uptake, even after adjusting for socioeconomic factors. These findings reinforce how caste operates not just as a social identity but as a determinant of health opportunity, intersecting with geography, gender and poverty to compound exclusion [40]. Previous studies also report similar findings [25]. Women belonging to the marginalised communities were reported to be 1.37 times more likely to lack access to healthcare services compared to men from the general category [41]. Religion, particularly among Muslim women, was associated with lower oral cancer screening uptake (aOR 0.68; 95% CI: 0.56–0.84), while Christians depicted the highest screening rates, followed by women following Hinduism. A systematic review by Kretzler
Education emerged as a strong enabler of screening, reinforcing the link between health literacy and the agency in health-seeking behavior. Women with no education had significantly lower odds of screening (aOR 0.66 (95% CI: 0.51–0.85)), with a clear gradient in screening rates across education levels. Illiterate women, who form a large proportion of rural, tribal and poorer groups, are often excluded from health communication strategies [42]. Another plausible explanation for low uptake is the misinterpretation of the screening question among women with low literacy. Although NFHS-5 interviews were conducted in local languages, studies show that women often confuse routine dental or oral examinations with cancer screening. In settings with limited prior exposure to cancer-related services, the concept of ‘screening’ itself may be unfamiliar, leading to underreporting or inconsistent responses [43]. This challenge has been noted in other surveys involving cancer screening, where women frequently misrecognised the nature or purpose of screening tests [44]. Such misclassification and misunderstanding may therefore partly contribute to the very low prevalence of self-reported oral cancer screening observed in our study.
The wealth gradient was consistently associated with screening uptake, with the richest quintile nearly twice as likely to be screened as the poorest, findings consistent with earlier work [3, 8, 20, 45]. Economic marginalisation intersects with other PROGRESS categories, particularly rural residence, low education and minority status, compounding social disadvantage. Given the persistent social gradient, policymakers must prioritise intersectional targeting rather than siloed outreach to single groups [20, 25, 35]. The data also suggested that female-headed households were associated with higher screening rates. In urban settings, 1.24% of female-headed households underwent screening compared to 0.81% in rural areas. This trend aligns with earlier research indicating that female-headed households were more likely to participate in breast and cervical cancer screening programs [20]. However, a contrasting study reported higher screening rates in male-headed households, underscoring the contradictory evidence from some studies, which reflects the complexity of gendered household decision-making in healthcare utilisation [20]. Media exposure played a significant role in widening the disparities across the socio-economic gradient, with rural women less likely to benefit from health promotion activities and campaigns. Previous studies also highlight the role of exposure to mass media in substantially increasing screening rates, particularly in urban areas [46, 47]. These findings underscore the need for inclusive communication strategies that account for disparities in access to digital and traditional media channels. Studies such as Changkun et al [20] show that women with higher education were significantly more likely to undergo oral cancer screening compared to those with no formal education in urban (aOR 1.29) as well as rural areas (aOR 1.24). Overall, education consistently enhances health literacy and screening uptake, although some variability across studies suggests that contextual differences may exist.
Regional disparities identified in the current analysis further underscore the inequitable implementation of oral cancer screening programs. The Southern region reported the highest screening uptake among women, across urban and rural areas, suggesting that cancer screening programs in these states may have been integrated into the primary health system, possibly benefiting from prior successful health campaigns and community health worker engagement. The findings align with Sen et al [35] who reported higher compliance with breast and cervical cancer screening in Tamil Nadu and Kerala, states known for their higher literacy levels and relatively stronger public health infrastructures. In contrast, the northeast and eastern regions exhibited alarmingly low coverage, with rural areas particularly underserved. Geospatial analysis reaffirmed these patterns. A significant Moran’s I statistic of 0.667 indicated strong spatial clustering of screening coverage, with hotspots concentrated in Punjab, Jammu & Kashmir and Kerala, and cold spots identified in central, eastern and northeastern regions. These findings align with the analysis by Gopika et al [48], which noted the highest oral cancer screening uptake among women in the Andaman and Nicobar Islands (10.1%). These variations suggest that differences in programme intensity, community engagement and health-system readiness, rather than geography alone, drive regional performance [20]. However, the NFHS-5 lacks information on programme delivery, health-system readiness or contextual factors that might explain these differences, and we cannot determine why some regions perform better or worse. Future studies should examine these high- and low-performing settings to identify transferable lessons for strengthening oral cancer screening across India. Some states may have benefited from pilot initiatives, donor-supported programs or stronger health system capacity; however, such information is not captured in the NFHS dataset, which limits our ability to interpret regional variations [49, 50].
Women with obesity or with diabetes/hypertension had higher screening rates. While this differs from some international evidence linking obesity to lower screening, contextual differences in care-seeking and provider engagement may explain these patterns [51, 52]. A similar contrast was seen for tobacco users, who had lower screening rates, particularly in rural areas, and the results are comparable to sub-national studies specifically targeting oral cancer screening [53]. Despite being at higher risk, lower screening uptake can be explained by several plausible reasons. Tobacco-using women often experience stigma, self-blame and fear of judgment by healthcare providers, which discourages them from oral examinations [54, 55]. Various forms of tobacco use among women are frequently normalised within households, reducing perceived susceptibility and need for screening [56]. Health-system barriers such as limited counselling, lack of women-centred tobacco cessation support and inadequate provider engagement further limit uptake [57–59].
Strengthening screening would therefore require a gender-responsive approach. Community-based studies demonstrate that integrating visual oral examinations into routine primary care, engaging frontline healthcare workers for household-level motivation, and utilising patient-navigation systems can substantially enhance early detection. Indian evidence consistently demonstrates that screen-positive women are far more likely to complete diagnostic confirmation and initiate treatment when accompanied by a patient navigator or a trusted community member [60]. Such ‘known faces’ help address fear, stigma, low autonomy and logistical barriers that commonly prevent women from following through with referrals. Experiences from breast and cervical cancer programmes in Tamil Nadu and Kerala confirm that navigator-supported pathways reduce loss to follow-up and improve treatment compliance. Tailored communication and culturally sensitive counselling further enhance uptake and linkage to care.
The study has several strengths and some limitations that warrant consideration
In conclusion, the poor uptake of oral cancer screening among Indian women reflects deeper governance and systemic issues. National and subnational level policies must embrace decentralised and equity-responsive strategies, prioritising regions with low baseline coverage and high risk of oral cancer. Geographically embedded disparities also necessitate region- or state-specific investments, including mobile outreach, culturally appropriate health promotion campaigns and capacity building of healthcare workers in the identification and reporting of oral cancers in women. Addressing the identified inequities and ensuring diversity and inclusion in program implementation will be key to oral cancer control in India.
Conflicts of interest
Authors have no conflicts of interest to declare.
Funding
No funding or financial grant was received to conduct the present study.
Authors’ contribution
NK, PH, YK and SR contributed to the conception and design of the study. NK, PH, GSJ and GB contributed to the analysis plan. PH, YK and SR contributed to the analysis and interpretation of data. NK, PH and GSJ drafted the manuscript. RS, ST and MG helped write the manuscript’s discussion. All authors reviewed the manuscript. All authors have approved the final version of the manuscript for submission.
Ethics
The NFHS-5 received ethical approval from the International Institute for Population Sciences (IIPS), Mumbai (2019–21). It was also reviewed by the ICF International Review Board (IRB), which approved it ethically. After receiving complete information about the goal and methodology of the survey, the respondents signed to confirm their agreement. Interviews were conducted only after receiving each participant’s informed consent.
Acknowledgments
Nil.
Data availability
The study utilises data from a published summary of the NFHS-5, which is publicly accessible and can be obtained by registering at https://dhsprogram.com/Countries/CountryMain.cfm?ctry_id=57&c=India. The corresponding author can provide the processed data upon reasonable request.
Consent for publication
Not applicable.
Clinical trial number
Not applicable (this is a cross-sectional study).
References
1. Kocarnik JM, Compton K, and Dean FE, et al (2022) Cancer incidence, mortality, years of life lost, years lived with disability, and disability-adjusted life years for 29 cancer groups from 2010 to 2019 JAMA Oncol 8(3) 420 PMCID: 8719276
2. Ferrari AJ, Santomauro DF, and Aali A, et al (2024) Global incidence, prevalence, years lived with disability (YLDs), disability-adjusted life-years (DALYs), and healthy life expectancy (HALE) for 371 diseases and injuries in 204 countries and territories and 811 subnational locations, 1990–2021: a systema Lancet 403(10440) 2133–2161 https://doi.org/10.1016/S0140-6736(24)00757-8
3. Johnson S, McDonald JT, and Corsten M (2012) Oral cancer screening and socioeconomic status J Otolaryngol Head Neck Surg 41(2) 102–107 PMID: 22569010
4. Bray F, Laversanne M, and Sung H, et al (2024) Global cancer statistics 2022: gLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries CA Cancer J For Clinicians 74(3) 229–263
5. Coelho KR (2012) Challenges of the oral cancer burden in India J Cancer Epidemiol 2012 701932 https://doi.org/10.1155/2012/701932 PMID: 23093961 PMCID: 3471448
6. Ferlay J, Colombet M, and Soerjomataram I, et al (2021) Cancer statistics for the year 2020: an overview Int J Cancer 2021
7. Mathur P, Sathishkumar K, and Das P, et al (2025) Global burden of 292 causes of death in 204 countries and territories and 660 subnational locations , 1990 – 2023 : a systematic analysis for the Global Burden of Disease Study 2023 Summary Background JAMA Netw Open 8(8) e2527805 PMID: 40833697 PMCID: 12368690
8. Dhane AS (2024) Understanding oral cancer disparities in India: exploring socioeconomic and demographic factors impacting access to care and outcomes Oral Oncol Rep 10 100441 https://doi.org/10.1016/j.oor.2024.100441
9. Gupta B, Bray F, and Kumar N, et al (2017) Associations between oral hygiene habits, diet, tobacco and alcohol and risk of oral cancer: a case–control study from India Cancer Epidemiol 51 7–14 https://doi.org/10.1016/j.canep.2017.09.003 PMID: 28968558
10. Kretzler B, König HH, and Brandt L, et al (2022) Religious denomination, religiosity, religious attendance, and cancer prevention. A systematic review Risk Manag Healthc Policy 15 45–58 https://doi.org/10.2147/RMHP.S341085 PMID: 35079226 PMCID: 8777031
11. Sankaranarayanan R, Ramadas K, and Amarasinghe H, et al (2015) Oral cancer: prevention, early detection, and treatment Disease Control Priorities, Third Edition (Volume 3): Cancer (The World Bank) p 85 https://doi.org/10.1596/978-1-4648-0349-9_ch5
12. Ministry of Health and Family Welfare; Government of India (2016) Operational framework on the managment of common cancers [Internet] Vol 4 [https://mohfw.gov.in/sites/default/files/Operational Framework Management of Common Cancers_1.pdf] Date accessed: 26/03/25
13. Sankaranarayanan R, Ramadas K, and Thara S, et al (2013) Long term effect of visual screening on oral cancer incidence and mortality in a randomized trial in Kerala, India Oral Oncol 49(4) 314–321 https://doi.org/10.1016/j.oraloncology.2012.11.004
14. International Institute for Population Sciences (2021) National family health survey (NFHS-5) India 2019-21 [Internet]. Demographic and health surveys pp 1–714 [https://dhsprogram.com/methodology/survey/survey-display-541.cfm] Date accessed: 30/08/23
15. Global Adult Tobacco Survey (GATS) (2017) Public use data file, India 2017 (Mumbai)
16. Tata Institute of Social Sciences, Ministry of Health and Family Welfare Government of India (2010) Global adult tobacco survey India 2009-2010 [Internet] pp 289 [https://www.who.int/tobacco/surveillance/survey/gats/ind/en/] Date accessed: 23/11/20
17. Goyal LD, Verma M, and Garg P, et al (2022) Variations in the patterns of tobacco usage among Indian females - findings from the global adult tobacco survey India BMC Womens Health 22(1) 442 https://doi.org/10.1186/s12905-022-02014-3 PMID: 36368987 PMCID: 9652978
18. Kumar A (2023) The transformation of The Indian Healthcare System Cureus 15(5) e39079 PMID: 37378105 PMCID: 10292032
19. Mishra GA, Shaikh HA, and Pimple SA, et al (2021) Determinants of compliance to population-based oral cancer screening program among low socioeconomic women in Mumbai, India Indian J Community Med 46(2) 210–215 PMID: 34321728 PMCID: 8281837
20. Changkun Z, Bishwajit G, and Ji L, et al (2022) Sociodemographic correlates of cervix, breast and oral cancer screening among Indian women PLoS One 17(5) 265881 https://doi.org/10.1371/journal.pone.0265881
21. The Demographic Health Survey Program (2020) DHS survey design: frequently asked questions what factors determine sample size? [Internet] pp 1–2 [https://dhsprogram.com/pubs/pdf/SAR15/SAR15.pdf] Date accessed: 11/11/24
22. The Demographic Health Surveys Program (2024) DHS methodology [Internet] [https://dhsprogram.com/methodology/survey-types/DHS-Methodology.cfm] Date accessed: 11/11/24
23. Ministry of Health and Family Welfare; Govt. of India (2016) Population based screening of common non-communicable diseases [Internet]. Training module for staff nurses [https://ab-hwc.nhp.gov.in/download/document/829bf86e304132dfae9962855c57dcd8.pdf] Date accessed: 11/11/24
24. Garg P, Krishnamoorthy Y, and Halder P, et al (2025) Urban-rural disparities in cervical cancer screening among Indian women between 30–49 years: a geospatial and decomposition analysis using a nationally representative survey BMC Cancer 25(1) 67 https://doi.org/10.1186/s12885-025-13446-z PMID: 39799327 PMCID: 11724505
25. Banerjee S (2021) Determinants of rural-urban differential in healthcare utilization among the elderly population in India BMC Public Health 21(1) 939 https://doi.org/10.1186/s12889-021-10773-1 PMID: 34001026 PMCID: 8130530
26. Shruti T, Khanna D, and Khan A, et al (2023) Status and determinants of early detection of oral premalignant and malignant lesions in India Cancer Control 30 https://doi.org/10.1177/10732748231159556 PMID: 36809192 PMCID: 9947682
27. Res M, Centre N, Aids N, et al (2012) Validity of self-reported morbidity Indian J Med Res 136(5) 722–724
28. Tugwell P, Petticrew M, and Kristjansson E, et al (2010) Assessing equity in systematic reviews: realising the recommendations of the commission on social determinants of health BMJ 341 c4739 https://doi.org/10.1136/bmj.c4739 PMID: 20837575
29. Anselin L, Syabri I, and Kho Y (2006) GeoDa : an introduction to spatial data analysis Geogr Anal 38(1) 5–22 https://doi.org/10.1111/j.0016-7363.2005.00671.x
30. Powers D, Yoshioka H, and Yun MS (2011) Mvdcmp: multivariate decomposition for nonlinear response models Stata J 11(4) 556–576 https://doi.org/10.1177/1536867X1201100404
31. Karran EL, Cashin AG, and Barker T, et al (2023) Using PROGRESS-plus to identify current approaches to the collection and reporting of equity-relevant data: a scoping review J Clin Epidemiol 163 70–88 https://doi.org/10.1016/j.jclinepi.2023.09.017 PMID: 37802205
32. Mayo M, Potugari B, and Bzeih R, et al (2021) Cancer screening during the COVID-19 pandemic: a systematic review and meta-analysis Mayo Clin Proc Innov Qual Outcomes 5(6) 1109–1117 https://doi.org/10.1016/j.mayocpiqo.2021.10.003 PMID: 34693211 PMCID: 8520861
33. Luciani S, Caixeta R, and Chavez C, et al (2023) What is the NCD service capacity and disruptions due to COVID-19? Results from the WHO non-communicable disease country capacity survey in the Americas region BMJ Open 13(3) 70085 https://doi.org/10.1136/bmjopen-2022-070085
34. United Nations (2021) Tackling inequalities in public service coverage to “build forward better” for the rural poor - policy brief [Internet] [https://unsceb.org/sites/default/files/2021-12/HLCP ITT Policy Brief Rural Inequalities 2021.pdf] Date accessed: 26/11/25
35. Sen S, Khan PK, and Wadasadawala T, et al (2022) Socio-economic and regional variation in breast and cervical cancer screening among Indian women of reproductive age: a study from National Family Health Survey, 2019-21 BMC Cancer 22(1) 1279 https://doi.org/10.1186/s12885-022-10387-9 PMID: 36476339 PMCID: 9727878
36. Sinha A, Kumar Sedai A, and Bahadur Rahut D, et al (2024) Well-being costs of unpaid care: gendered evidence from a contextualized time-use survey in India World Dev 173 106419 https://doi.org/10.1016/j.worlddev.2023.106419
37. Kundra S, Sreen N, and Dwivedi R (2023) Impact of work from home and family support on Indian women’s work productivity during COVID-19 Vikalpa J Decis Makers 48(1) 39–53 https://doi.org/10.1177/02560909231162918
38. Singh SK, Chauhan K, and Puri P (2023) Chronic non-communicable disease burden among reproductive-age women in India: evidence from recent demographic and health survey BMC Women’s Health 23(1) 20 https://doi.org/10.1186/s12905-023-02171-z PMID: 36650531 PMCID: 9843940
39. Idris IB, Hamis AA, and Bukhori ABM, et al (2023) Women’s autonomy in healthcare decision making: a systematic review BMC Womens Health 23(1) 643 https://doi.org/10.1186/s12905-023-02792-4 PMID: 38042837 PMCID: 10693143
40. Thapa R, Van Teijlingen E, and Regmi PR, et al (2021) Caste exclusion and health discrimination in South Asia: a systematic review Asia Pacific J Public Heal 33(8) 828–838 https://doi.org/10.1177/10105395211014648
41. Ahmed S and Mahapatro S (2023) Inequality in healthcare access at the intersection of caste and gender Contemp Voice Dalit 15(1_suppl) S75–S85 https://doi.org/10.1177/2455328X221142692
42. Siva N, Mohanty K, and Rath R, et al (2025) A systematic scoping review of health-seeking behavior and healthcare utilization in tribal communities of Odisha, India: concentration on maternal and child health BMC Public Health 25(1) 2801 https://doi.org/10.1186/s12889-025-24079-z PMID: 40819166 PMCID: 12357398
43. Rogers ES, Wallace LS, and Weiss BD (2006) Misperceptions of medical understanding in low-literacy patients: implications for cancer prevention Cancer Control 13(3) 225–229 https://doi.org/10.1177/107327480601300311 PMID: 16885919
44. ecancer (2023) Survey reveals widespread confusion and misconceptions about ovarian cancer screening among a majority of women - ecancer [Internet] News [https://ecancer.org/en/news/23668-survey-reveals-widespread-confusion-and-misconceptions-about-ovarian-cancer-screening-among-a-majority-of-women] Date accessed: 26/11/25
45. Karanth S, Mistry S, and Wheeler M, et al (2024) Persistent poverty disparities in incidence and outcomes among oral and pharynx cancer patients Cancer Causes Control 35(7) 1063–1073 PMID: 38520565 PMCID: 11217118
46. Ismail AI, Jedele JM, and Lim S, et al (2012) A marketing campaign to promote screening for oral cancer J Am Dent Assoc 143(9) e57–e66 https://doi.org/10.14219/jada.archive.2012.0328 PMID: 22942156
47. Schliemann D, Su TT, and Paramasivam D, et al (2019) Effectiveness of mass and small media campaigns to improve cancer awareness and screening rates in Asia: a systematic review J Glob Oncol 5(5) 1–20 PMID: 30969807 PMCID: 6528732
48. Gopika MG, Prabhu PR, and Thulaseedharan JV (2022) Status of cancer screening in India: an alarm signal from the National Family Health Survey (NFHS-5) J Fam Med Prim Care 11(11) 7303–7307 https://doi.org/10.4103/jfmpc.jfmpc_1140_22
49. Ministry of Health and Family Welfar (2019) National programme for prevention and control of cancer, diabetes, cardiovascular diseases and stroke (NPCDCS) [Internet] [https://main.mohfw.gov.in/Major-Programmes/non-communicable-diseases-injury-trauma/Non-Communicable-Disease-II/National-Programme-for-Prevention-and-Control-of-Cancer-Diabetes-Cardiovascular-diseases-and-Stroke-NPCDCS] Date accessed: 17/10/22
50. Ministry of Health and Family Welfare (2023) National programme for prevention and control of non-communicable diseases (2023-2030) [Internet]. Operational guidelines [https://pib.gov.in/PressReleseDetail.aspx?PRID=1924730]
51. Bernard M, Löbner M, and Lordick F, et al (2022) Cancer prevention in females with and without obesity: does perceived and internalised weight bias determine cancer prevention behaviour? BMC Womens Health 22(1) 511 https://doi.org/10.1186/s12905-022-02085-2 PMID: 36494719 PMCID: 9733287
52. Urbute A, Kjaer SK, and Kesmodel US, et al (2022) Women with obesity participate less in cervical cancer screening and are more likely to have unsatisfactory smears: results from a nationwide Danish cohort study Prev Med (Baltim) 159 107072 https://doi.org/10.1016/j.ypmed.2022.107072
53. Menon I, Kumar P, and Rebello P, et al (2025) Oral cancer awareness and screening practices in urban versus rural Indian population: web-based survey Bioinformation 21(05) 1253–2126 https://doi.org/10.6026/973206300211253 PMID: 40822778 PMCID: 12357727
54. Goel S, Walia D, and Kumar R (2024) The hidden crisis: health impacts of tobacco and nicotine products on Indian women J Fam Med Prim Care 13(11) 4751–4754 https://doi.org/10.4103/jfmpc.jfmpc_1741_24
55. Kumar DV, Amandeep D, and Rathee DS, et al (2025) Barriers to treatment seeking among female smokers in rural community in north India; a qualitative study J Popul Ther Clin Pharmacol 32(3) 693–697
56. Jassem E (2014) The many faces of tobacco use among women Med Sci Monit 20 153–162 https://doi.org/10.12659/MSM.889796 PMID: 24487778 PMCID: 3915001
57. Sultana S, Inungu J, and Jahanfar S (2025) Barriers and facilitators of tobacco cessation interventions at the population and healthcare system levels: a systematic literature review Int J Environ Res Public Health 22(6) 825 https://doi.org/10.3390/ijerph22060825 PMID: 40566253 PMCID: 12193092
58. Pollak KI, Arredondo EM, and Yarnall KSH, et al (2001) How do residents prioritize smoking cessation for young ‘high-risk’ women? factors associated with addressing smoking cessation Prev Med (Baltim) 33(4) 292–299 https://doi.org/10.1006/pmed.2001.0884
59. Chaudhary J, Gupta E, and Singh PK, et al (2023) Tobacco exposure among antenatal women in India: challenges in tobacco screening & cessation counselling Indian J Med Res 158(5&6) 477–482 https://doi.org/10.4103/ijmr.ijmr_188_23 PMID: 38088423 PMCID: 10878484
60. Sharma P, Khanna D, Pradhan S, et al (2023) Community cancer screening at primary care level in Northern India: determinants and policy implications for cancer prevention Fam Med Community Heal 11(Suppl 1) e002397 https://doi.org/10.1136/fmch-2023-002397
61. Thakur J, Jeet G, and Nangia R, et al (2019) Non-communicable diseases risk factors and their determinants: a cross-sectional statewide STEPS survey, Haryana, North India PLoS One 14(11) 1–19 https://doi.org/10.1371/journal.pone.0208872
62. Thakur JS, Jeet G, and Pal A, et al (2016) Profile of risk factors for non-communicable diseases in Punjab, Northern India: results of a state-wide STEPS survey PLoS One 11(7) 1–16 https://doi.org/10.1371/journal.pone.0157705
63.Thakur J, Prinja S, and Jeet G, et al (2016) Costing of a state-wide population based cancer awareness and early detection campaign in a 2.67 million population of Punjab state in Northern India Asian Pacific J Cancer Prevention 17(2) 791–797 https://doi.org/10.7314/APJCP.2016.17.2.791
Supplementary Table S1. Distribution of various factors as per coverage of oral cancer screening among Indian women aged 30–49years.
Supplementary Table S2. Sub-national coverage of oral cancer screening among Indian women 15–49 years.