Background: Immune check point inhibitors (ICIs) have an established role in Microsatellite-Instability–High (MSI-H) and Combined Positive Score (CPS) high advanced gastric/gastroesophageal (G/GE) adenocarcinomas, but there is limited real world data with regard to practice patterns, and efficacy of standard doses (SD-ICIs) and alternative lower doses (LD-ICIs).
Methods: A retrospective study of patients with advanced G/GE adenocarcinomas receiving ICIs was conducted. The primary endpoint of the study was 12-month overall survival (OS), which was computed by Kaplan-Meier method.
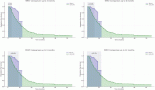
Results: A total of 91 patients were available for analysis during the study period. Seventy-four patients (81%) received nivolumab, while the remaining received pembrolizumab. Fifteen patients (16%) had MSI-H status and had a 12-month OS of 60% and median OS of 15 months (median follow-up – 38.3 months). In the Microsatellite-Stable (MSS) cohort (84%; n = 76), ICIs (combined with chemotherapy) were used predominantly in pre-treated patients (54%; n = 41). Patients with CPS ≥5 (72%; n = 55) had improved survival compared to patients with CPS <5 (28%; n = 21) (12-month OS: 52% vs. 26%; Median OS: 12.8 months vs. 3.2 months; p = 0.005). There was no difference in survival between patients who received SD-ICIs (54%; n = 41) and LD-ICIs (46%; n = 35) (12-month OS: 42% vs. 48%; Median OS: 8.7 months vs. 11 months; p = 0.44).
Conclusions: Patients with advanced G/GEJ adenocarcinomas in the real world predominantly received ICIs during later lines of therapy as opposed to first line therapy. Using a CPS cutoff of ≥5 as opposed to CPS <5 predicts for improved survivals in MSS patients and patients receiving low dose ICIs have similar survival outcomes to patients receiving standard dose ICIs within the confines of a heterogenous study cohort.